Table of Contents
QMS manager job or role is a common question when medical device companies start implementing ISO 13485 and defining responsibilities within their quality management system.
A QMS manager is usually a dedicated position with a critical role in maintaining regulatory compliance, so we recommend you to keep it separate! Sure, you’re a start-up, but as a medical device company, bite the bullet and hire a person for this Job.
Don’t make the Management Representative or a Unit Manager, the QMS Manager. This role is essential for keeping the focus on regulatory and standards compliance and quality assurance, blending other roles into it only dilutes the integrity of the position. You will be hard-pressed to claim that quality objective was met. It would only pander lip service to quality. More importantly, you would more than likely fail a QMS audit as a result.
Our recommendation is ” Don’t do it! ” it’s not worth the hassle.
QMS manager job or role: choosing a QMS Manager
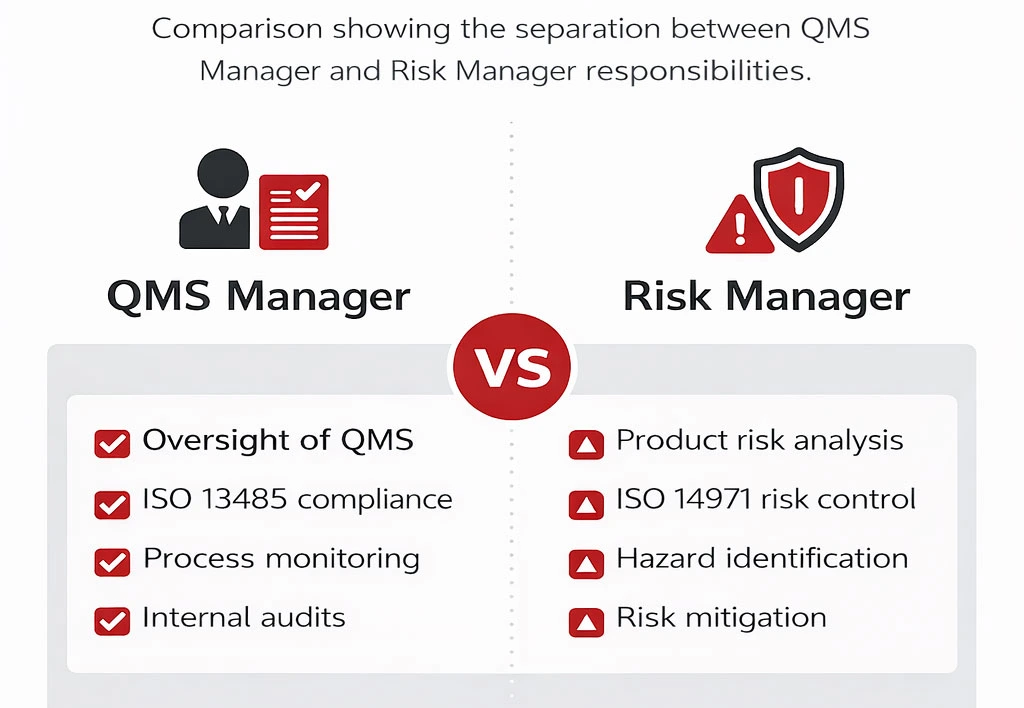
QMS Manager is responsible for developing the Quality strategies, policies, processes, standards and systems for the Company and they have a role to play and it’s not the same as the QMS Manager. The QMS Manager is not the HR Manager, not the Purchase Manager, not any other position or role. They are the final arbiter of oversight. Note: I did not say quality but of “oversight”. They determine if the proper process was properly followed. They cannot tell if the quality is there. They cannot validate that regulatory and standard compliance was achieved.
Rather, the quality should be there if the process was done correctly. i.e. the QMS Manager double checks.
In a more pragmatic way, a QMS manager has surveillance over the whole organization, the QMS processes, and tasks inside an organization and checks that everything is working and done correctly – and compliance is being respected.
Risk and QMS management: 2-in-1 or not?
The new ISO 13485:2016 states that anything you do that affects the quality system needs to be viewed from a risk perspective (risk-based approach). As such, many companies believe that the QMS Manager role can be combined with the Risk Manager role.
Although convenient at first glance, it’s not practical in any way and it will represent a huge problem for smaller companies. This is exactly where the qms manager job or role discussion becomes important, because separating responsibilities helps maintain proper oversight of quality processes and regulatory compliance.
Choosing a Risk Manager
Essentially a Risk Manager needs to understand, analyze and address the various risk issues to help their organizations detect potential sources of hazards or harm. They have to know and understand what risks are inherent in the medical device under development. They also have to know how to mitigate any risks and reduce the potential for harm.
A QMS manager doesn’t understand the product or its associated risk. In the qms manager job or role debate, this distinction is critical because the QMS Manager focuses on compliance oversight, while the Risk Manager focuses on product risk.
In this author’s opinion, the Unit Manager or Project manager should make the best Risk Manager for that project. A product manager could also step-in. The point is to assign this critical role or Risk Manager to someone who truly understands the medical device, product or service because they would have better insight into the nature of the risks associated with the use of that device than would a QMS manager.
Separate roles would mean that the Risk Manager can then interact with the QMS system and allow the QMS Manager to do their job correctly, ensuring QMS ‘Oversight’ of the Risk processes for that medical device/project.
In the Start-up or Small company, warm bodies are at a premium. We understand that. But the role of QMS manager is not simply quality, it is compliance and in a regulated industry, compliance is critical to market success. Fail in compliance, forget getting your product into the marketplace.
So bite the bullet, a QMS manager is for quality and compliance whereas the Risk Manager needs intimate product knowledge to address the potential for harm and harm mitigation.
Why the QMS Manager Job or Role Must Stay Independent
he qms manager job or role must remain independent in medical device companies to ensure objective oversight of quality processes and regulatory compliance. Separating this role from operational or risk management responsibilities helps maintain a reliable and audit-ready quality management system.
Reference:
http://www.allaboutcareers.com/careers/job-profile/risk-manager
Key Questions About the QMS Manager Job or Role in MedTech
Is a QMS Manager a job or just a role in a medical device company?
In most medical device companies, the QMS Manager should be a dedicated job rather than an additional role. The QMS manager job or role discussion often arises when companies try to combine responsibilities, but ISO 13485 compliance requires clear oversight of quality processes and regulatory requirements.
Why should the QMS Manager and Risk Manager roles be separated?
The QMS Manager is responsible for overseeing the quality management system, while the Risk Manager focuses on product risk analysis and mitigation under ISO 14971. Combining these roles can create conflicts of interest and weaken independent oversight of risk management activities.
Can the QMS Manager also act as a Risk Manager?
Although some small companies attempt to combine the roles, it is generally not recommended. The QMS Manager focuses on compliance and process oversight, while the Risk Manager requires deep understanding of the medical device, its hazards, and risk controls.
Who should typically be responsible for risk management in a MedTech company?
Risk management is usually best handled by someone directly involved in product development, such as a project manager, product manager, or engineering lead. These roles understand the device, its intended use, and potential hazards better than a QMS Manager.
Why is the QMS Manager role important for ISO 13485 compliance?
The QMS Manager ensures that processes are followed, documentation is maintained, and audits can demonstrate compliance with ISO 13485. Without clear oversight, companies risk regulatory findings, failed audits, or delays in bringing medical devices to market.




