CAPA & Non-Conformance — Driven by Events, Not Assumptions
Every issue starts as an event.
The system helps you decide what it becomes.
In qmsWrapper, CAPA and Non-Conformance are not standalone tools.
They are part of an event-driven quality system that captures issues early and escalates them only when it makes sense.
Not every issue is a CAPA.
But every issue must be controlled.
One entry point.
No blind spots.
Quality issues don’t start as CAPAs or non-conformities.
They start as signals noticed during real work.
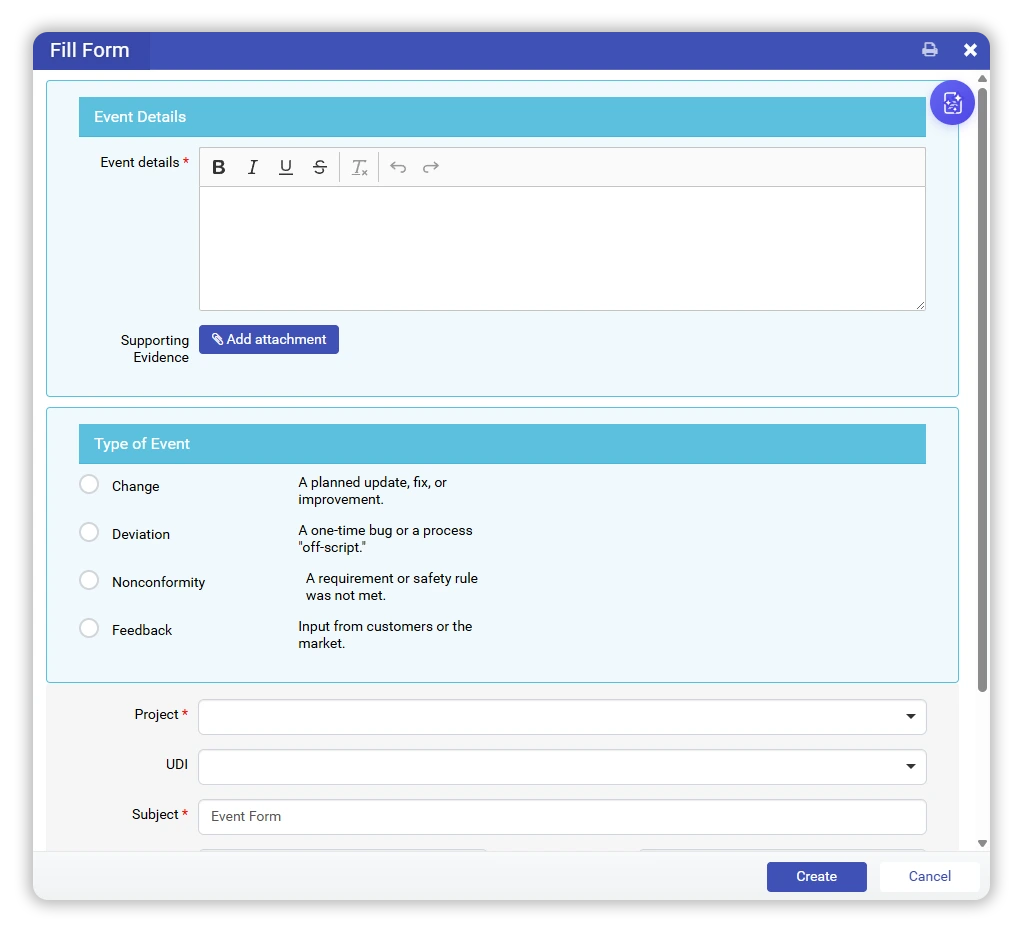
In qmsWrapper, every signal is captured as a Quality Event — simple to log, structured from the start, and never informal.
From that moment on, the issue is visible, traceable, and assessable.
If it isn’t captured as an event,
it isn’t under control.
CAPA and Non-Conformance: Classification Before Escalation
Once an event is logged, the system supports structured classification.
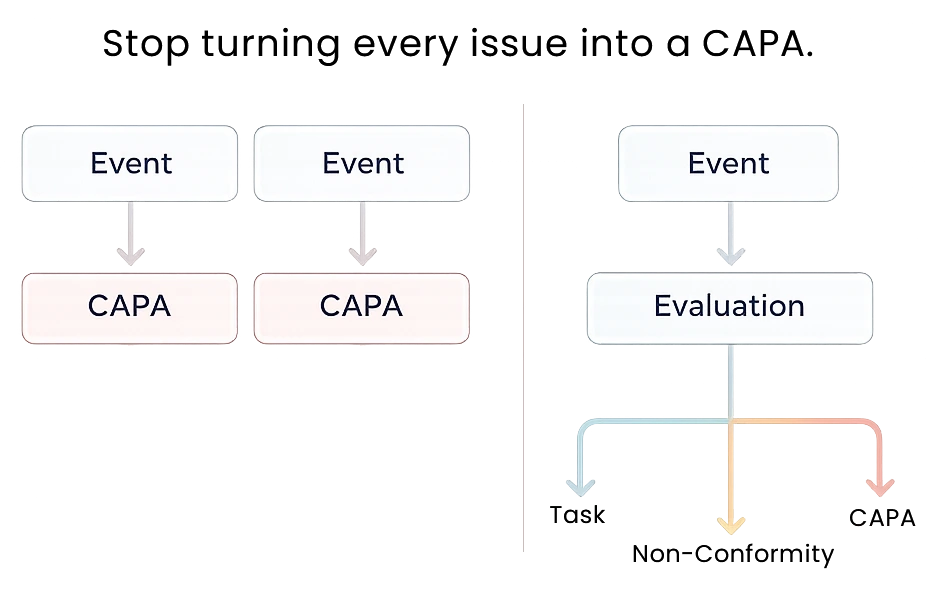
Some events become Non-Conformances.
Some remain deviations or are resolved with simple actions.
Some are documented and closed.
Non-Conformance is treated as a core QMS object, not just a step on the way to CAPA.
This keeps daily quality work clean, focused, and defensible.
Risk decides, not habit
Only when analysis shows a systemic or recurring issue does escalation continue.
qmsWrapper supports risk-based triage so that:
- minor issues don’t inflate CAPA logs
- serious problems get the attention they deserve
- decisions are proportional and explainable
For QMS managers, this means fewer CAPAs — and stronger ones.
CAPA is not a starting point.
It’s a decision.
Clear oversight, audit-ready by design
Non-Conformances and CAPAs each have their own dedicated modules, with clean logs designed for everyday QMS management.
AI assists by:
- carrying data from events into NCs and CAPAs
- reducing manual re-typing
- keeping investigations and actions consistent
When a CAPA is opened, it runs as a closed-loop process — from investigation to effectiveness — fully linked back to the original event.
Auditors see not just what happened, but why decisions were made.
See how CAPA and Non-Conformance works in an event-driven system
Your QMS shouldn’t turn every issue into a CAPA —
and it shouldn’t lose control of the ones that aren’t.
Let us show you how events, non-conformances, and CAPAs work together in one coherent system.
How CAPA and Non-Conformance Actually Work in Practice
What is the difference between CAPA and Non-Conformance in a QMS?
A Non-Conformance records a specific instance where a requirement was not met.
CAPA is a structured process used when the issue is systemic, recurring, or high-risk.
Not every Non-Conformance requires a CAPA — but every CAPA should be traceable back to its originating event.
Should every issue be escalated to CAPA?
No. Escalating every issue to CAPA leads to overloaded systems and weak investigations.
In a risk-based QMS, only issues with significant impact or recurrence are escalated.
Other issues can be handled as tasks, deviations, or standalone Non-Conformances.
What triggers a CAPA in an event-driven QMS?
A CAPA is triggered when analysis shows:
- a systemic issue
- recurring problems
- significant risk to product or compliance
In qmsWrapper, this decision is based on structured event evaluation — not habit or manual judgment alone.
How does an event-driven QMS improve CAPA management?
An event-driven QMS captures issues at the moment they occur and structures them as Quality Events.
From there, the system:
- classifies the issue
- evaluates risk
- determines the appropriate action
This ensures CAPAs are created only when justified, improving both efficiency and audit readiness.
How are CAPAs linked to Non-Conformances and events?
CAPAs are not isolated records.
Each CAPA is linked back to:
- the original event
- related Non-Conformances
- investigation data and actions
This creates full traceability from detection to resolution — a key requirement for audits.
What does a closed-loop CAPA process mean?
A closed-loop CAPA process ensures that every issue goes through:
- investigation
- root cause analysis
- corrective actions
- effectiveness verification
The process is only complete when effectiveness is confirmed and linked back to the original event.
How does AI support CAPA and Non-Conformance workflows?
AI assists by:
- transferring data from events into CAPAs and Non-Conformances
- reducing manual data entry
- maintaining consistency across records
This improves speed, accuracy, and audit readiness without replacing human decision-making.
Why is traceability important for CAPA and Non-Conformance?
Auditors don’t just check if actions were taken — they verify why decisions were made.
Traceability ensures that every CAPA and Non-Conformance is:
- linked to its source
- supported by data
- fully explainable
This turns compliance from documentation into defensible evidence.
