Pre-Validated SaaS eQMS Platform
Reduce your CSV workload — without compromising regulatory defensibility.
Pre-validated, version-controlled SaaS platform delivered with comprehensive vendor-side validation documentation.
✔ Faster implementation
✔ Reduced Computer System Validation (CSV) effort
✔ Impact-based re-validation for new releases
✔ Built for ISO 13485 & FDA-regulated environments

Delivered with Structured Vendor-Side Validation
qmsWrapper is a Pre-Validated SaaS platform, version-controlled and designed to streamline both initial and ongoing CSV while maintaining full regulatory defensibility.
Unlike traditional QMS platforms that require extensive initial configuration and heavy re-validation cycles, qmsWrapper significantly reduces initial validation effort and enables impact-based re-validation with each software update — eliminating unnecessary full-system re-validation.
Platform Highlights
- Trusted by regulated MedTech and SaMD organizations
- Pre-validated SaaS platform
- Reduced ongoing re-validation effort
- Aligned with GAMP 5 principles
- Risk reduction in regulatory compliance
Why This Matters for Your Organization
Designed to Minimize Validation Friction While Maintaining Full Regulatory Defensibility
Implementation & Validation Efficiency
Reduced validation effort
Delivered with comprehensive vendor-side validation documentation, significantly lowering the workload required for CSV.
Faster implementation & time-to-value
Operate within days — not months — using a pre-validated, structured SaaS environment.
Structured customer acceptance model
Dedicated validation execution template enabling documented review, customer-side testing, and formal system approval.
Regulatory Confidence & Documentation Depth
Audit-ready documentation structure
Version-controlled validation reports, requirements specifications and structured release documentation.
Risk-based validation approach
Each system version undergoes internal risk-based validation aligned with GAMP 5 principles and industry best practices for configurable off-the-shelf (COTS) software.
Regulatory alignment by design
Supports ISO 13485, FDA QMSR / 21 CFR Part 820, 21 CFR Part 11, and EU MDR expectations.
Full traceability & change transparency
Clear linkage between system requirements, use cases, validation testing, and release updates.
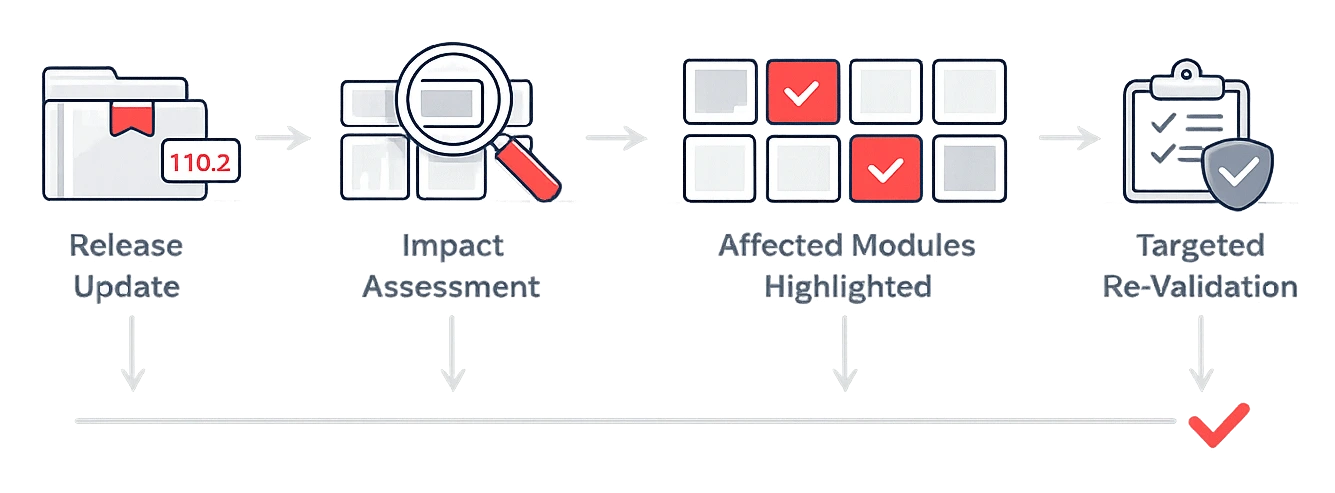
Controlled Release
& Re-Validation Management
- Predictable release impact assessment
Clear identification of affected functional areas for controlled re-validation planning. - Reduced ongoing CSV workload for new releases
Focus validation activities only on impacted areas rather than re-validating the entire system after each update.
Regulatory Framework Alignment
The qmsWrapper validation model aligns with:
- ISO 13485
- FDA QMSR / 21 CFR Part 820
- 21 CFR Part 11 (ERES)
- EU MDR
In accordance with regulatory expectations, final validation and acceptance remain the responsibility of the customer for their intended use.

Vendor-Side Validation Documentation
For each system release, qmsWrapper provides version-controlled validation documentation, including:
- ERES Compliance Statement
- ERES Rationale
- Validation Summary
- Validation Support Statement
- System Requirements Specification
- Use Cases & Functional Traceability
- Validation Report (per system version)
- Release Notes (per system version)
This documentation demonstrates structured testing, traceability, and change control across system versions.
Customer Validation Execution Support
Customers are supported through:
- Customer Validation Execution & Acceptance Form
This structured template aligns directly with system functionality and enables:
- Review of vendor validation documentation
- Execution of customer-side validation activities
- Formal system acceptance documentation


Industry-Proven Validation Model
This validation approach reflects established industry best practices for regulated SaaS platforms and configurable off-the-shelf (COTS) software.
Developed by leadership with over 30 years of experience in regulated quality and compliance environments, qmsWrapper supports medical device and SaMD organizations operating under ISO 13485, FDA, and EU MDR frameworks.
Access to Validation Documentation
The full validation documentation package is available to contracted customers. Prospective customers may request a validation overview under appropriate confidentiality arrangements.
Validation Clarity: What MedTech Teams Need to Know
What does “Pre-Validated SaaS” actually mean for a medical device company?
A Pre-Validated SaaS platform provides structured vendor-side validation documentation for each system release. This significantly reduces the customer’s Computer System Validation (CSV) workload while maintaining regulatory defensibility under ISO 13485, FDA QMSR, and EU MDR expectations. The customer remains responsible for final validation and intended use acceptance.
Does a Pre-Validated SaaS eliminate the need for customer validation?
No. Regulatory expectations require that the customer performs final validation for their intended use. However, with structured vendor documentation (Validation Report, SRS, ERES statements, Release Notes), the validation effort is focused and risk-based rather than full-system re-validation.
How does impact-based re-validation work in a SaaS eQMS?
Each system release undergoes internal risk-based validation aligned with GAMP 5 principles. Release documentation clearly identifies affected functional areas, allowing customers to validate only impacted elements instead of re-validating the entire system after every update.
What validation documentation is provided with each release?
For every system version, qmsWrapper provides:
- ERES Compliance Statement
- ERES Rationale
- Validation Summary
- Validation Support Statement
- System Requirements Specification
- Use Cases & Functional Traceability
- Validation Report (per system version)
- Release Notes (per system version)
- Customer Validation Execution & Acceptance Form
This documentation ensures structured testing, traceability, and transparent change control across versions.
How does this support ISO 13485 and FDA QMSR compliance?
The validation model aligns with:
- ISO 13485 quality system requirements
- FDA QMSR / 21 CFR Part 820
- 21 CFR Part 11 (ERES)
- EU MDR expectations
The structured documentation model supports audit readiness, traceability, and risk-based validation practices consistent with regulatory guidance.
Is this suitable for SMEs or only large enterprises?
The Pre-Validated SaaS model is particularly beneficial for MedTech startups and growing SMEs. It reduces implementation time, lowers validation overhead, and avoids the complexity and cost associated with enterprise-scale customization and repeated re-validation cycles.
How does vendor-side validation reduce ongoing CSV workload?
By providing version-controlled validation documentation and structured release impact assessment, the system allows customers to focus only on affected functional areas. This reduces repetitive validation effort and supports controlled, predictable re-validation management.
Can prospective customers review validation documentation before purchase?
A validation overview can be shared under appropriate confidentiality arrangements. Full validation documentation packages are available to contracted customers.
