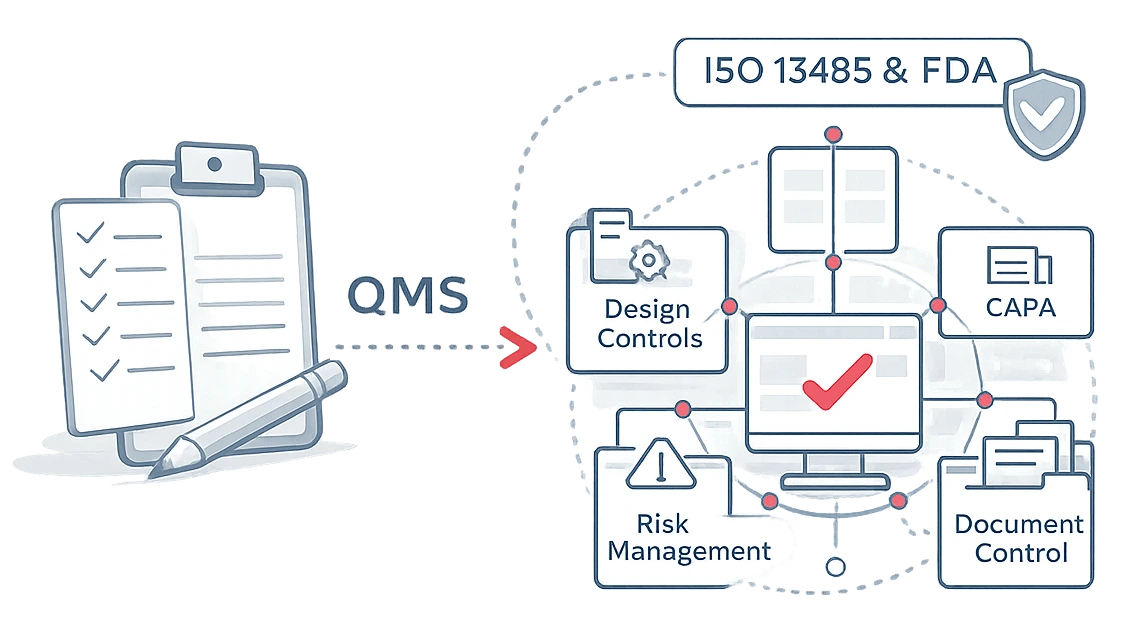
ISO 13485 & FDA Compliance Built Into Your QMS
qmsWrapper is designed specifically for regulated MedTech environments. Instead of forcing you to configure compliance from scratch, the system comes with pre-configured workflows, templates, and structures aligned to the regulations auditors and notified bodies actually expect to see — across design, risk, quality events, and documentation.
Compliance shouldn’t be assembled.
It should already be there.
Standards and Regulations qmsWrapper Supports
Quality & Risk Standards
- ISO 13485 – Medical Devices QMS
- ISO 14971 – Risk Management
- ISO 9001 – Quality Management
- IEC 62304 – Software Lifecycle
- IEC 60601 – Medical Electrical Equipment
Regulatory Frameworks
- EU MDR
- EU IVDR
- UK MDR
- EU MDR Annex II & III (Technical Documentation)
- FDA 510(k) readiness
Data, Security & Records
- FDA 21 CFR Part 820 (QMSR-aligned)
- FDA 21 CFR Part 11 (eRecords & eSignatures)
- SOC 2
- ISO 27001
- HIPAA
- GDPR
How ISO 13485 & FDA Compliance Is Structured in qmsWrapper
qmsWrapper reflects real audit logic. Quality processes, design controls, risk management, and technical documentation are already organized the way auditors and notified bodies review them — with clear structure, traceability, and evidence paths.
You don’t “map” compliance later. You work inside it from day one.
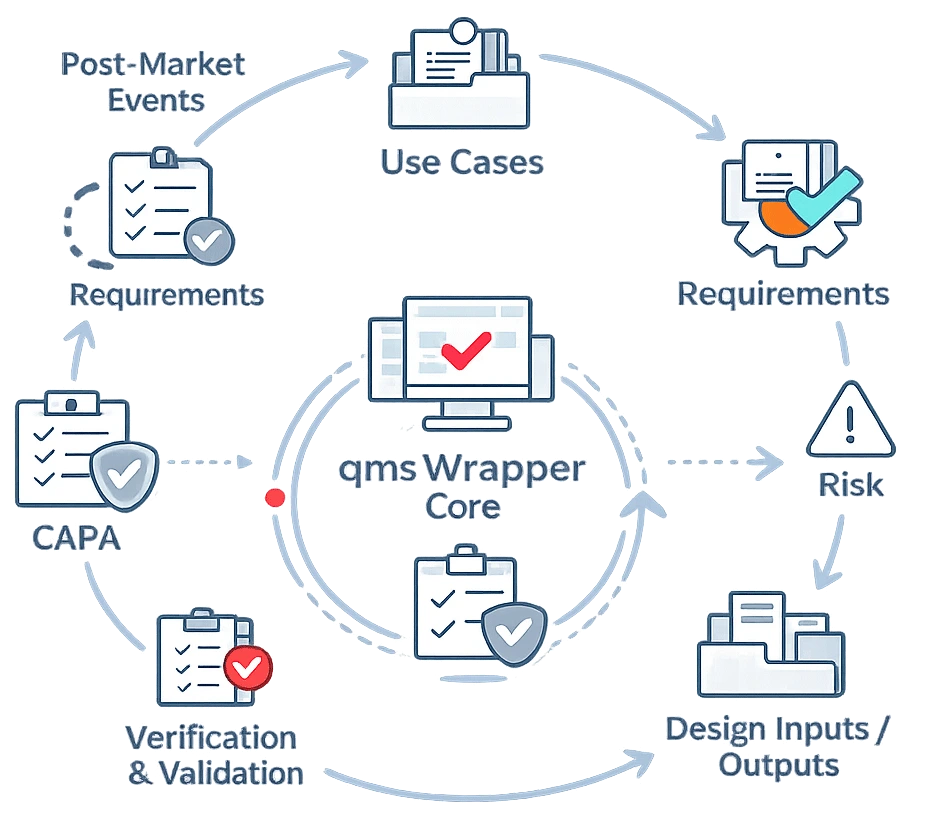
From Design to Post-Market — One Aligned System
- Design History File (DHF) and Technical File structure aligned to MDR and FDA expectations
- Traceability Matrix linking use cases, requirements, risks, tests, and changes
- Risk management embedded across design and quality events
- CAPA, Change Control, Deviations, Complaints, and Nonconformity fully connected
- Electronic records and approvals ready for Part 11 environments
Everything is linked. Nothing lives in isolation.d to the relevant quality process, ensuring nothing is missed, delayed, or handled outside the system.
When something changes,
compliance updates with it.
qmsWrapper
Built for Regulatory Shifts — Not Static
Compliance
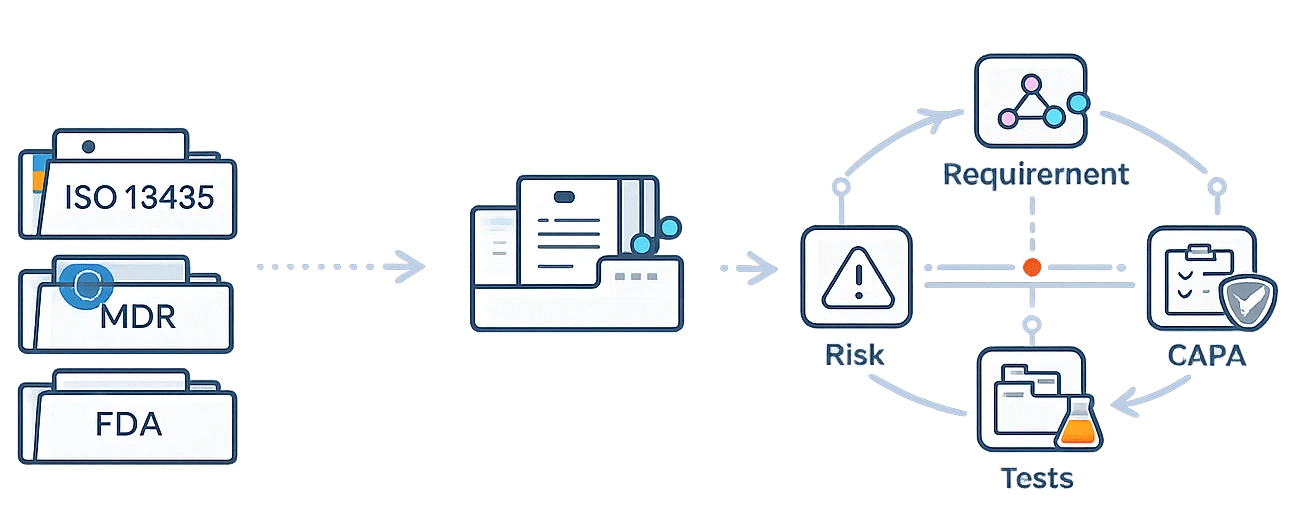
Regulations evolve. Your system shouldn’t collapse every time they do.
qmsWrapper is structured around controlled Change Control and connected traceability. When requirements change — whether ISO updates, MDR guidance, or FDA QMSR — you adapt within the system, not outside it.
No rebuild.
No broken links.
No traceability gaps.
Suitable for:
- Teams preparing for certification
- Companies expanding between EU and FDA frameworks
- Growing MedTech organizations scaling product portfolios
Ready to See How This Works in Practice?
See how qmsWrapper supports your specific regulatory scope — and how quickly you can be audit-ready without overengineering your QMS.
Regulatory Clarity: What ISO 13485 & FDA Compliance Really Means in Practice
What does ISO 13485 & FDA Compliance mean for a QMS platform?
ISO 13485 & FDA Compliance means the QMS is structured to support design controls, risk management, document control, CAPA, and audit traceability in alignment with ISO 13485:2016 and FDA 21 CFR Part 820 requirements. In qmsWrapper, these structures are pre-configured rather than manually built.
Does qmsWrapper support FDA 21 CFR Part 820 and Part 11?
Yes. qmsWrapper supports FDA 21 CFR Part 820 (QMSR-aligned) processes including design controls, CAPA, and record management. It also supports Part 11 requirements for electronic records and electronic signatures through controlled workflows and approval tracking.
How is ISO 14971 risk management connected to ISO 13485 compliance?
ISO 14971 risk management is integrated directly into design and quality processes. Risks are linked to requirements, design inputs/outputs, verification tests, and quality events, ensuring continuous traceability required under ISO 13485 and FDA expectations.
Is EU MDR technical documentation supported within the same system?
Yes. The Design History File (DHF) and Technical File structure are aligned with EU MDR Annex II and III requirements. Traceability between requirements, risks, tests, and post-market events supports lifecycle documentation expectations under MDR.
Can the system adapt to regulatory updates such as FDA QMSR transition?
Yes. qmsWrapper is built with configurable workflows and form-driven logic, allowing adaptation to evolving regulatory frameworks such as FDA’s transition toward ISO 13485 alignment under QMSR, without breaking traceability.
Is this suitable for both startups and scaling MedTech companies?
Yes. Startups preparing for first certification, SMEs entering new markets, and companies transitioning between EU MDR and FDA frameworks can use the same structured system without rebuilding compliance architecture.
How ISO 13485 and FDA QMSR compliance map across qmsWrapper
qmsWrapper is pre-configured for ISO 13485:2016 and the FDA QMSR (21 CFR Part 820 as amended, effective 2 February 2026, incorporating ISO 13485:2016). Coverage includes:
- ISO 13485:2016 §7.3 design and development controls, including §7.3.10 design files.
- FDA design controls under 21 CFR 820.30, now read through the FDA QMSR.
- ISO 13485:2016 §5.6 (management review), §8.2.3 (internal audit), and §8.2.4 (monitoring).
- FDA 21 CFR 820.20 (management responsibility) and §820.22 (quality audit).
- CE marking under MDR Annex II and III with Notified Body routes Annex IX, X, or XI, and UKCA under UK MDR 2002.
