Three Levels of Wrapper Capabilities. Migrate when ready.
Start with the processes you need today. Expand into QMS control. Add Vigilance when evidence, impact and regulatory change need to stay connected.
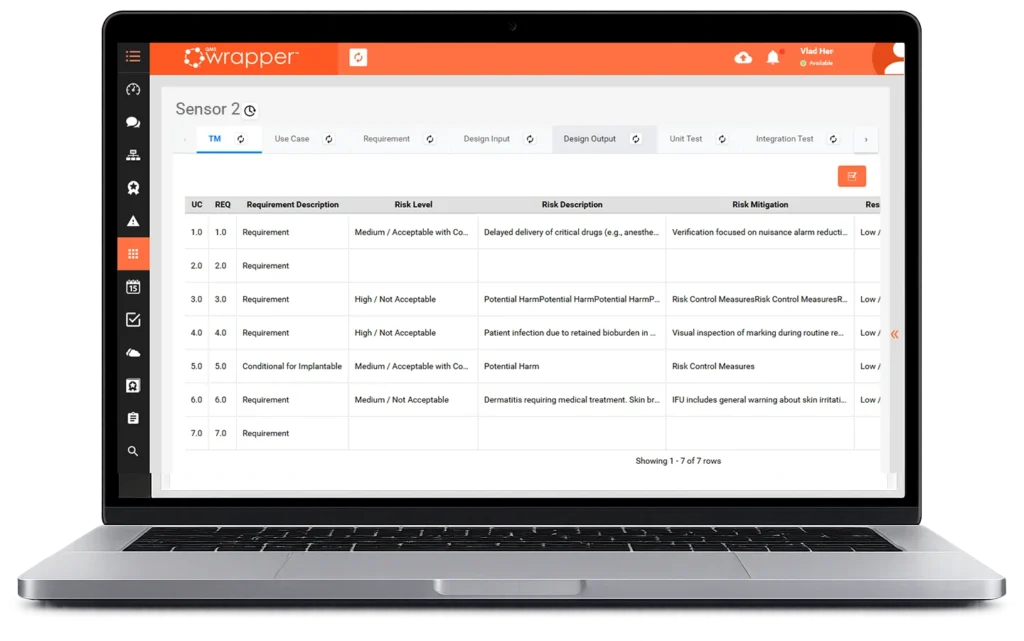
Wrapper Process
From paper to controlled processes
- Paper forms to eForms
- Approvals and records
- Form builder + process editor
- QMS Manual framework
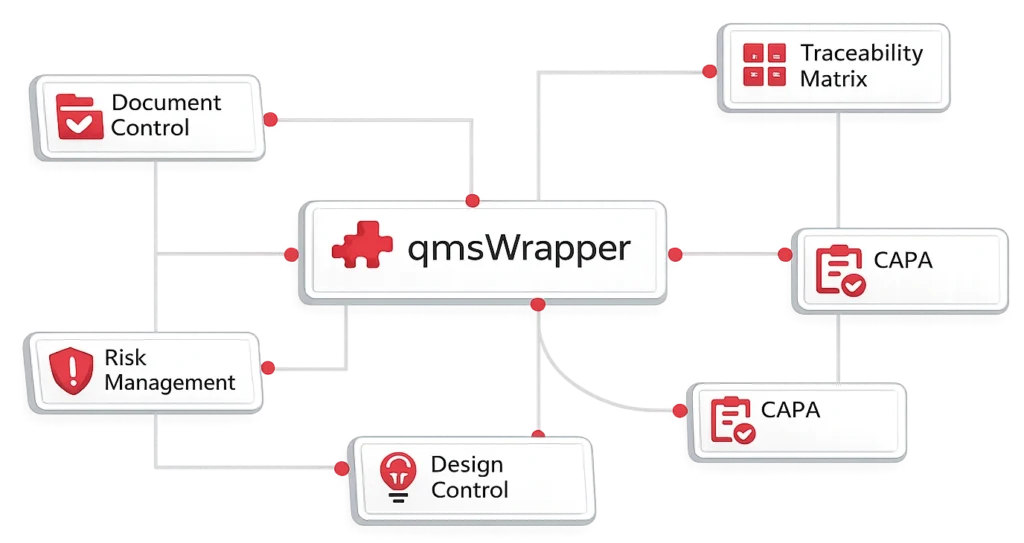
Wrapper QMS
From processes to connected QMS
- Event intake and triage
- CAPA, Change, N-C, Deviation
- Risk Module + Hazard Log
- Training, suppliers, feedback
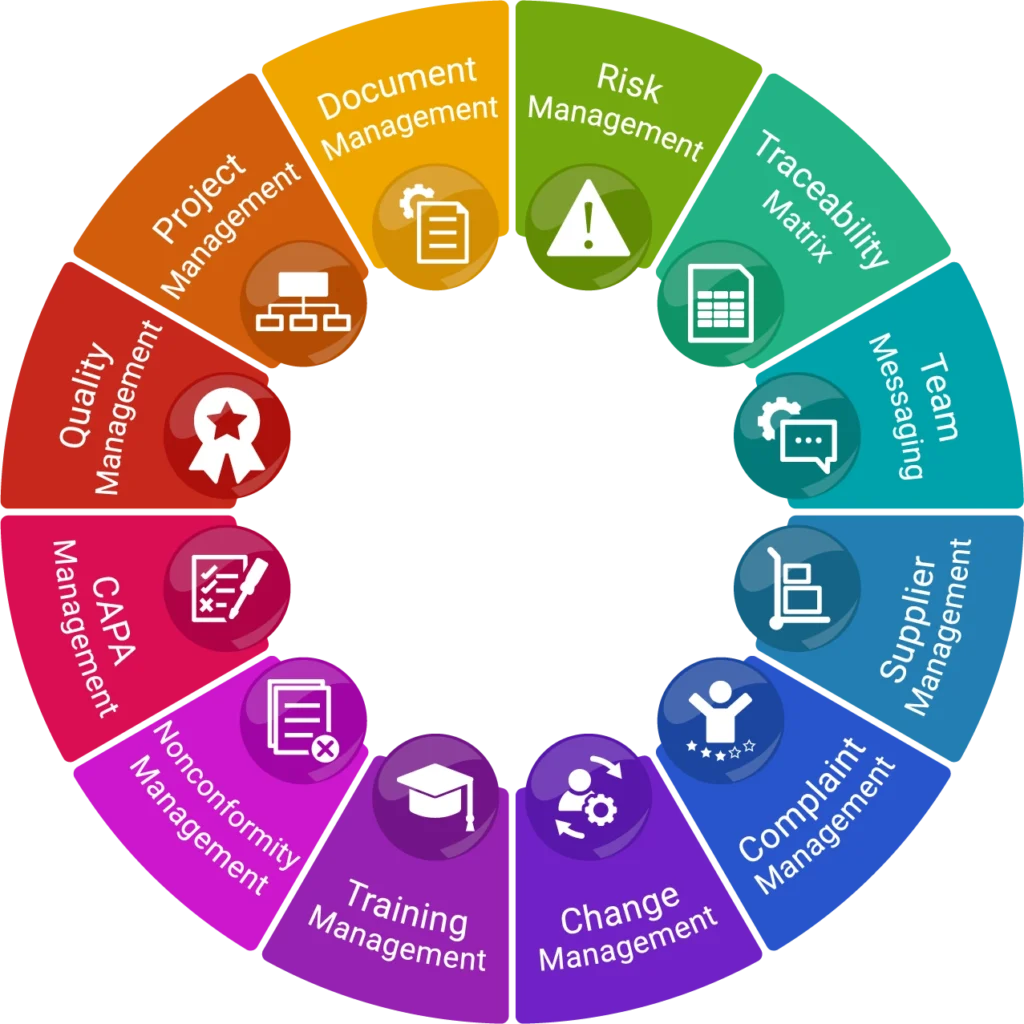
Wrapper Vigilance
From QMS to audit-ready evidence
- Event-to-Evidence AI
- TF/TM impact mapping
- PMS and audit evidence
- Evidence-ready
Built for Compliance