ISO 13485 Change and Deviation Management
Event-driven workflows for controlled changes, temporary deviations, and audit-ready traceability.
A small process adjustment.
A quick fix on the shop floor.
A document update “just this once.”
In regulated MedTech environments, uncontrolled change is how traceability breaks, risks go out of date, and audit findings begin.
qmsWrapper ensures every Change and Deviation is formally captured, assessed, approved, and connected to the right records before implementation starts.
Most audit problems start long before the audit
Many Change and Deviation issues begin as small operational decisions:
Over time, these “small exceptions” create larger compliance gaps:
By the time an auditor asks questions, teams are often forced to reconstruct what happened manually.
That reconstruction is slow, stressful, and difficult to defend.
If it wasn’t logged, it wasn’t controlled.
Change control begins before approval
Changes and deviations do not begin as formal approvals.
They begin the moment someone proposes doing something differently.
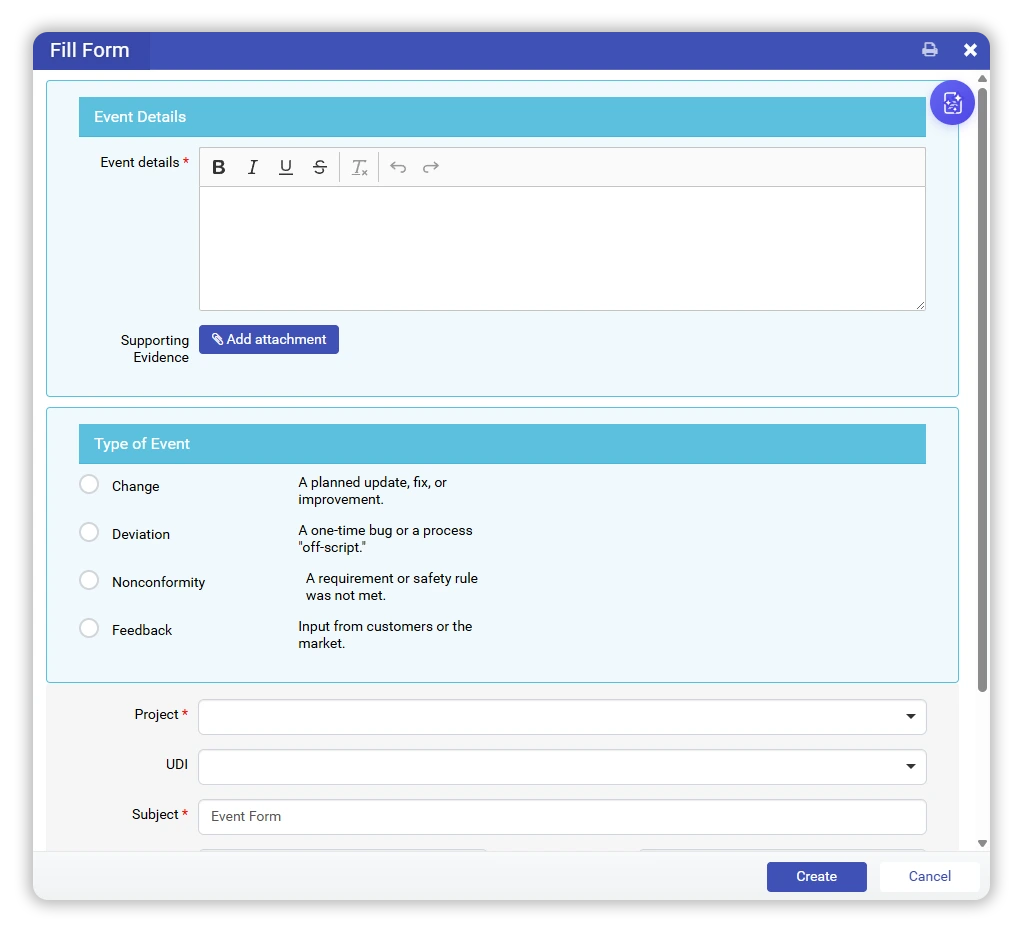
In qmsWrapper, that moment is captured immediately as an Event — clearly classified as:
- Change
- Deviation
- Non-Conformance
- Feedback
This prevents uncontrolled decisions from entering the system informally and ensures every action begins with operational context.
Temporary deviations should not become permanent habits
qmsWrapper helps MedTech teams clearly separate:
Deviations
Controlled temporary departures from approved processes or documentation.
Changes
Permanent updates to products, processes, specifications, or controlled documents.
This distinction matters because many audit findings begin when temporary actions quietly become permanent operational behavior without proper review.
The system keeps decisions deliberate, risk-based, and fully traceable.

Every change carries downstream impact
A process update rarely affects only one document.
qmsWrapper helps teams maintain visibility across these relationships before implementation happens.
This reduces the risk of:
Change control is not documentation overhead.
It is operational governance.
Context follows the workflow
AI assists by carrying information forward from the original Event into related Change or Deviation workflows.
This helps teams:
- reduce repetitive data entry
- preserve operational context
- maintain connected records
- keep affected requirements and risks visible
- accelerate review and approval cycles
Instead of manually reconstructing relationships later, teams maintain continuity from the first signal through final implementation.
Less administrative overhead.
More informed decisions.
Fewer disconnected changes.
Built for real audit questions
For QMS managers, qmsWrapper provides immediate visibility into every operational decision connected to Change and Deviation workflows.
For auditors, this creates a clean operational trail across Events, Changes, Risks, CAPAs, and Technical File history — without manual reconstruction.
Connected workflows. Traceable decisions. Audit-ready history.Auditors follow relationships, not isolated documents.
Change Control Questions MedTech Teams Ask
What is the difference between a Change and a Deviation in ISO 13485?
A Deviation is a temporary departure from an approved process or requirement. A Change is a permanent modification to controlled processes, products, or documentation.
Why is undocumented change a compliance risk?
Undocumented changes can break traceability, invalidate risk assessments, and create gaps between actual operations and approved procedures.
When should a change trigger risk re-evaluation?
Risk should be re-evaluated whenever a change affects product requirements, processes, suppliers, verification activities, or intended use.
How does FDA QMSR affect change management?
FDA QMSR strengthens alignment with ISO 13485 risk-based quality management principles, increasing focus on traceability, documented decisions, and controlled implementation.
Why do auditors review change history?
Auditors use change history to verify that companies maintain control over risk, approvals, implementation, and downstream product impact.
Controlled change should not require manual reconstruction
qmsWrapper helps MedTech teams manage Change and Deviation workflows with connected Events, risk visibility, and full lifecycle traceability.
See how real teams maintain audit-ready change control without disconnected spreadsheets, isolated approvals, or missing context.
