AI QMS for Medical Devices
Understand what must change — before you act.
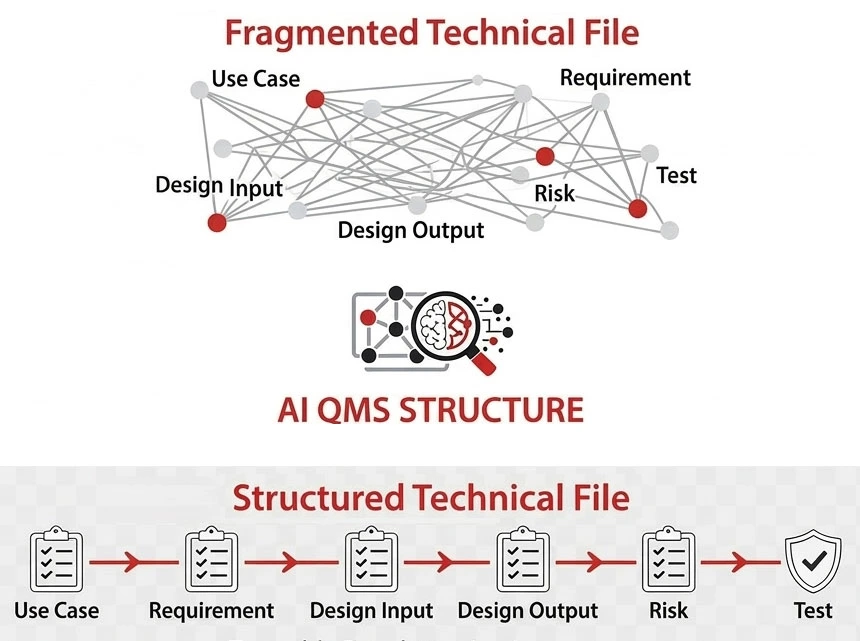
Most QMS systems document change.
Few help you understand its impact across requirements, risk, and the Technical File.
This report explains how operational AI changes that.
Table of Contents
Why AI Is Becoming Critical for Medical Device QMS
Medical device regulations don’t just require documentation.
They require:
- traceability across the Technical File
- documented change impact analysis
- clear connections between risk, requirements, and verification
Most teams still manage this manually.
The Real Problem Isn’t Documentation
A single change can affect:
- multiple requirements
- risk assessments
- verification activities
- documentation
But most QMS systems don’t show these connections.
They leave teams to figure it out manually.
The real challenge is understanding what must change when something changes.
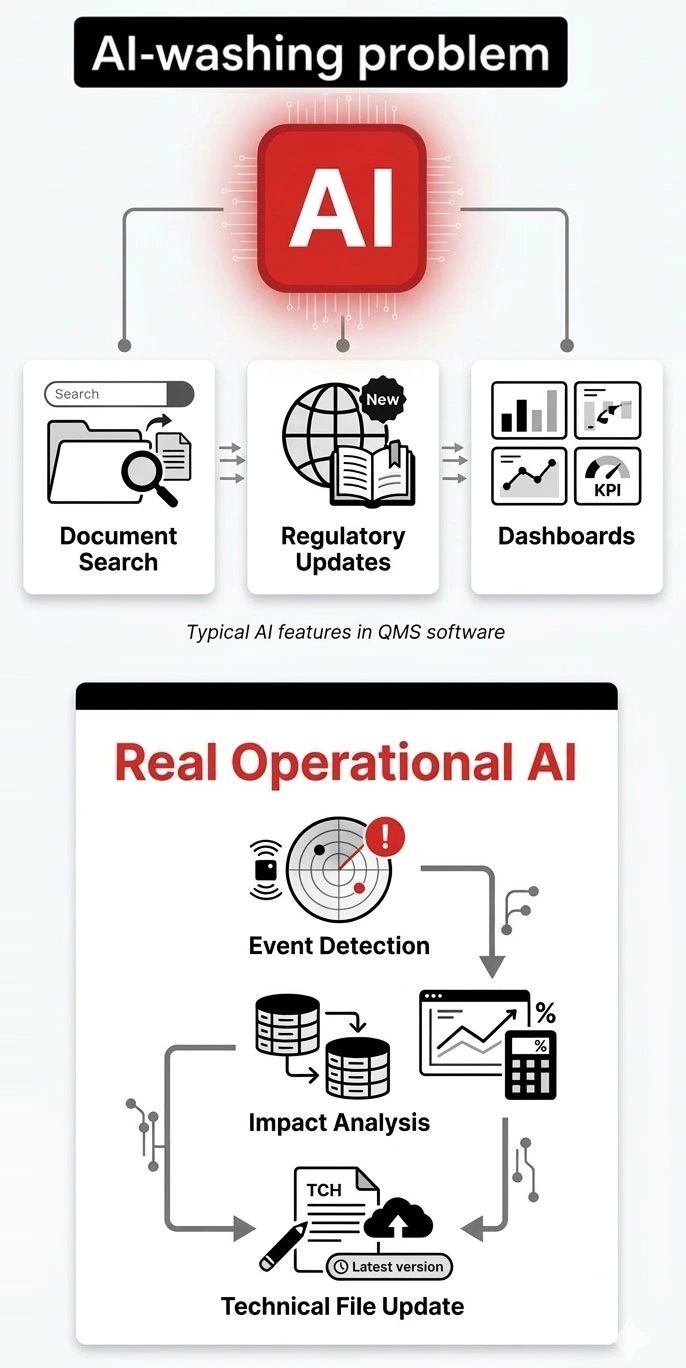

AI in QMS Is Often Misunderstood
Most platforms offer:
- dashboards
- search
- regulatory updates
This is observational AI.
It helps you see data — but not manage change.
Operational AI Changes the Workflow
Instead of analyzing data after the fact,
operational AI works inside the QMS.
It helps:
- evaluate events
- analyze change impact
- update the Technical File
- maintain traceability


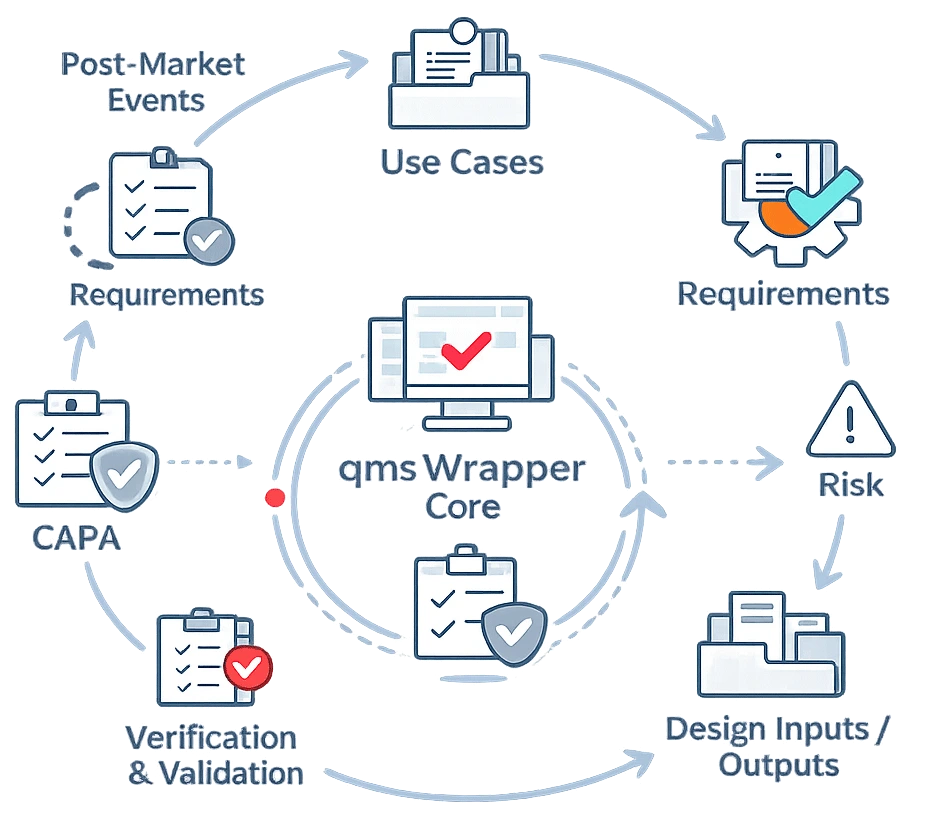
What You Will Learn in the Report
- why most “AI in QMS” is surface-level
- what real change impact analysis actually requires
- how operational AI supports traceability in practice
- how modern QMS systems connect risk, requirements, and testing
- how hybrid AI (SQL + semantic search) works in real workflows
Download the AI QMS Report
Learn how operational AI supports medical device compliance and audit readiness.
The report explores:
- AI architecture in modern QMS systems
- the role of change impact analysis
- how search intelligence supports regulatory traceability
Common Questions About AI QMS for Medical Devices
What is an AI QMS for medical devices?
An AI QMS integrates artificial intelligence into quality workflows such as event evaluation, change impact analysis, and technical documentation management.
How does AI help medical device compliance?
AI can assist with identifying relationships between events, requirements, risks, and verification evidence, helping teams maintain regulatory traceability.
What makes operational AI different?
Operational AI supports the compliance workflow itself, helping teams evaluate events and update technical documentation rather than simply retrieving documents.
Why is change impact analysis important?
Regulators expect companies to demonstrate how design changes affect risk, testing, and technical documentation. Modern QMS systems help identify and manage this impact.