ISO 14971 Risk Management Built for Real-World MedTech Teams
Structured Hazard Logs. Automatic re-evaluation. Full lifecycle traceability.
qmsWrapper supports ISO 14971–aligned risk management by keeping hazards, hazardous situations, and risk evaluations in live Hazard Logs, connected to real design and quality work.
When something changes, risk comes back into focus automatically — without extra effort.
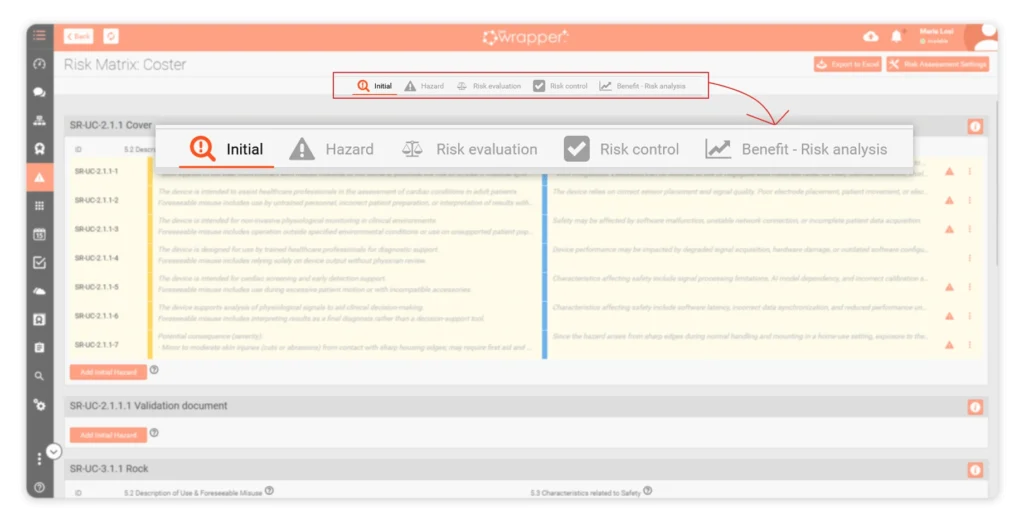
ISO 14971 Risk Management with Live Hazard Logs
Instead of managing risk in disconnected files, Hazard Logs keep all risk-related decisions in one place.
They evolve as your product evolves — without rebuilding the Risk Management File each time.
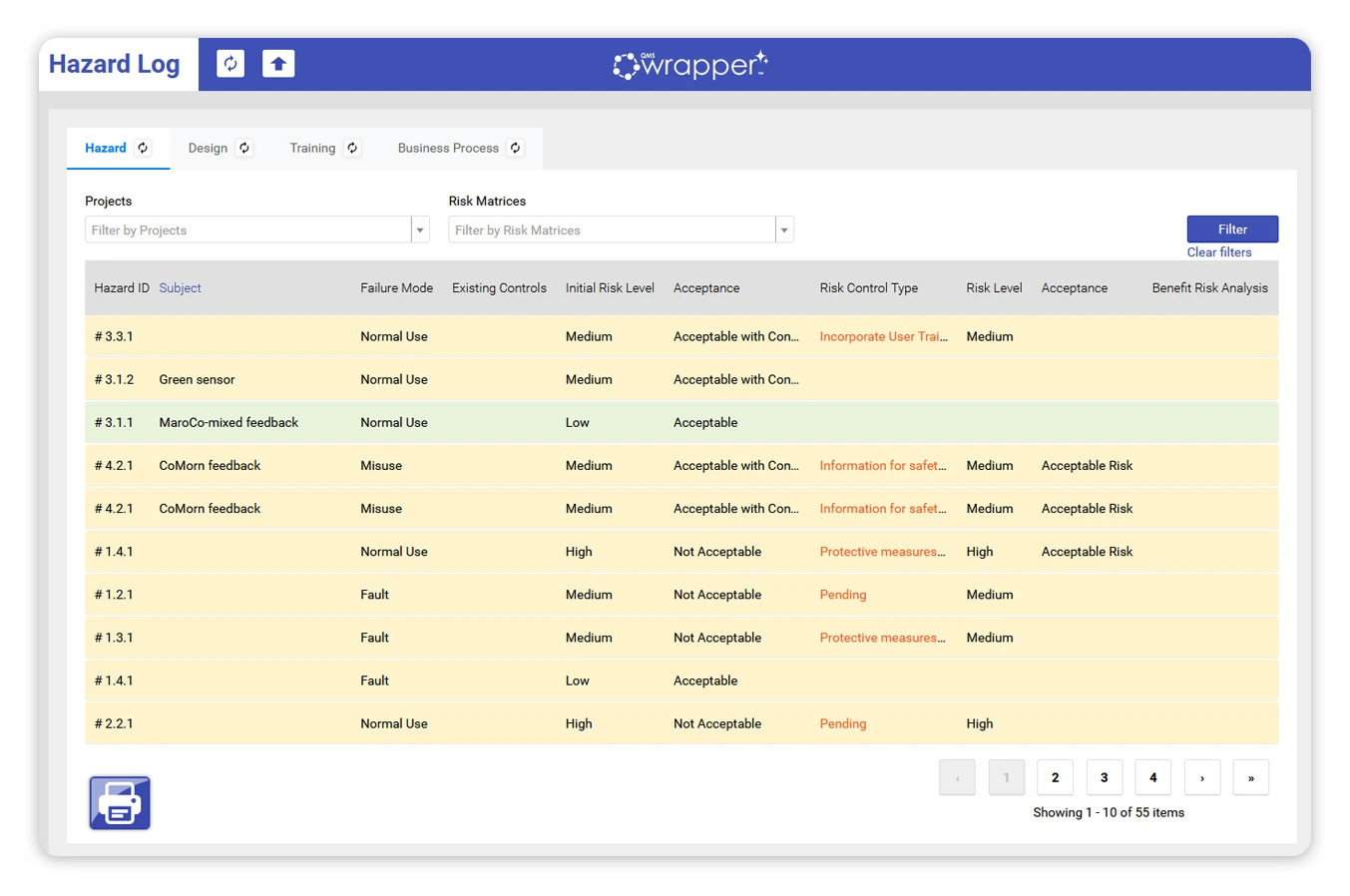
When risk stays static, teams are forced
to remember what the system should remember.
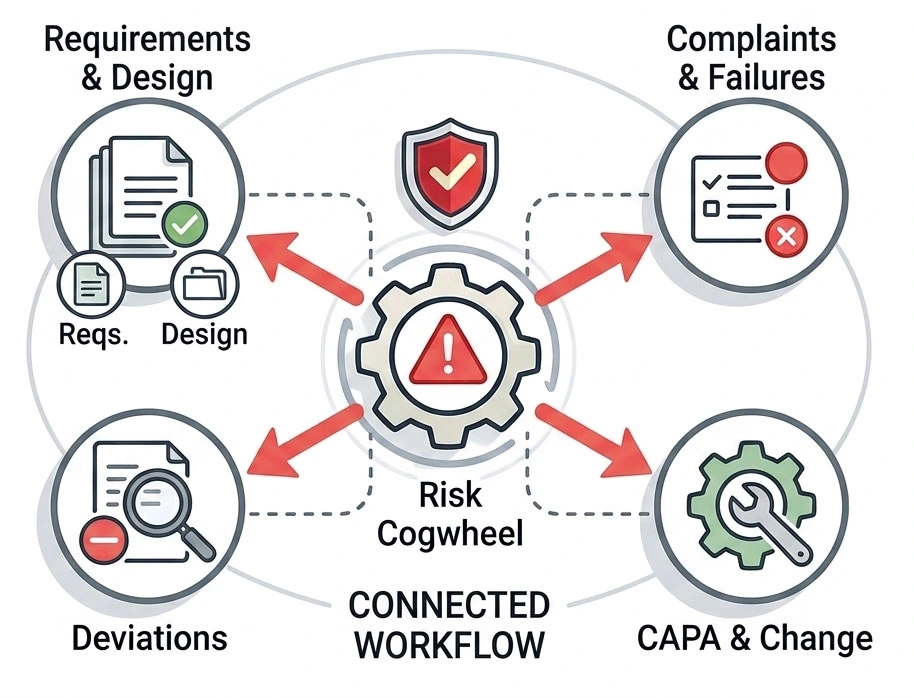
Risk stays connected to everyday quality work
Risk is not treated as a separate activity.
It stays connected to what teams already work on every day.
This means risks are directly linked to:
- requirements and design elements
- complaints, failures, and deviations
- CAPA and Change activities
When new information appears, affected risks are visible immediately — no manual cross-checking.
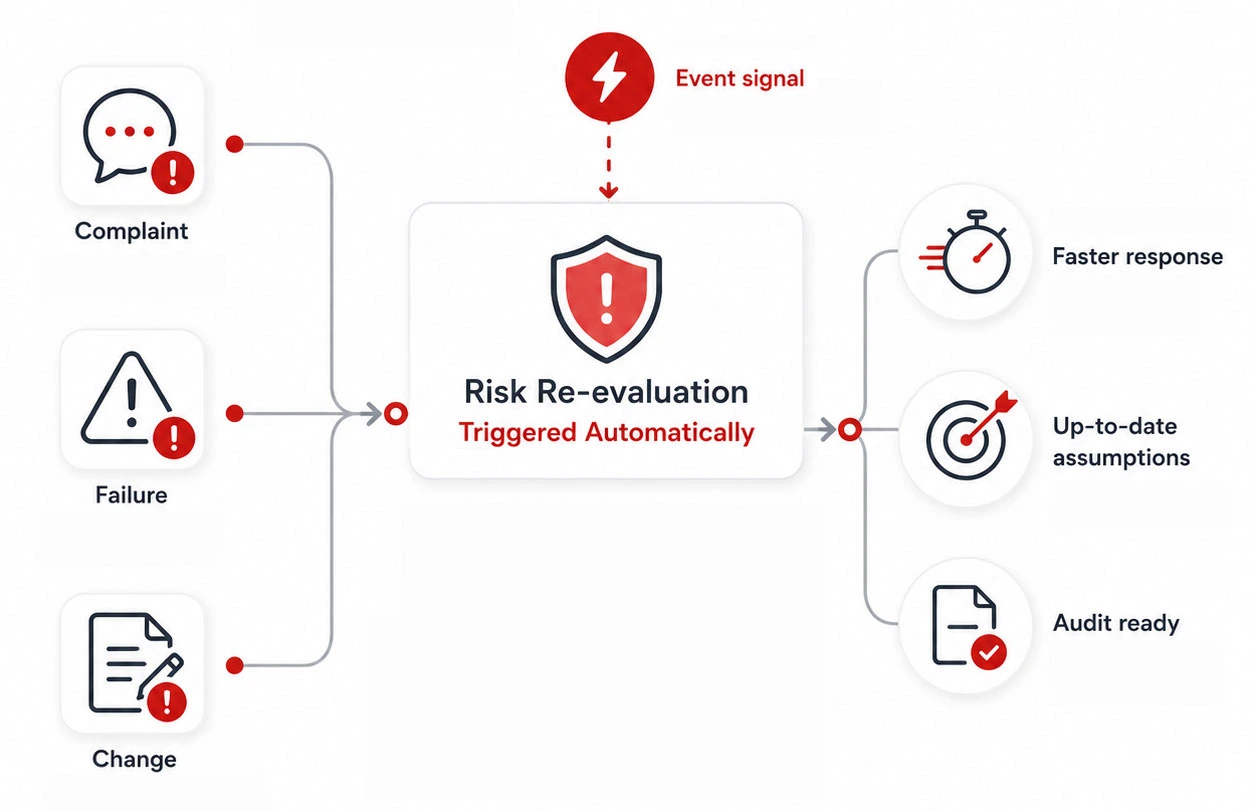
Risk-based thinking works best when it follows real events, not calendars.
Event-driven
risk re-evaluation
Instead of relying on periodic reviews, risk is revisited when it matters most.
Complaints, failures, or changes can trigger risk re-evaluation automatically, helping teams:
- react faster to new evidence
- avoid forgotten or outdated risk assumptions
- reduce last-minute audit preparation
This aligns naturally with ISO 14971 principles and modern regulatory expectations, including FDA’s move toward system-based, risk-focused oversight (QMSR).
Less chasing documents.
More confidence that risks are under control.
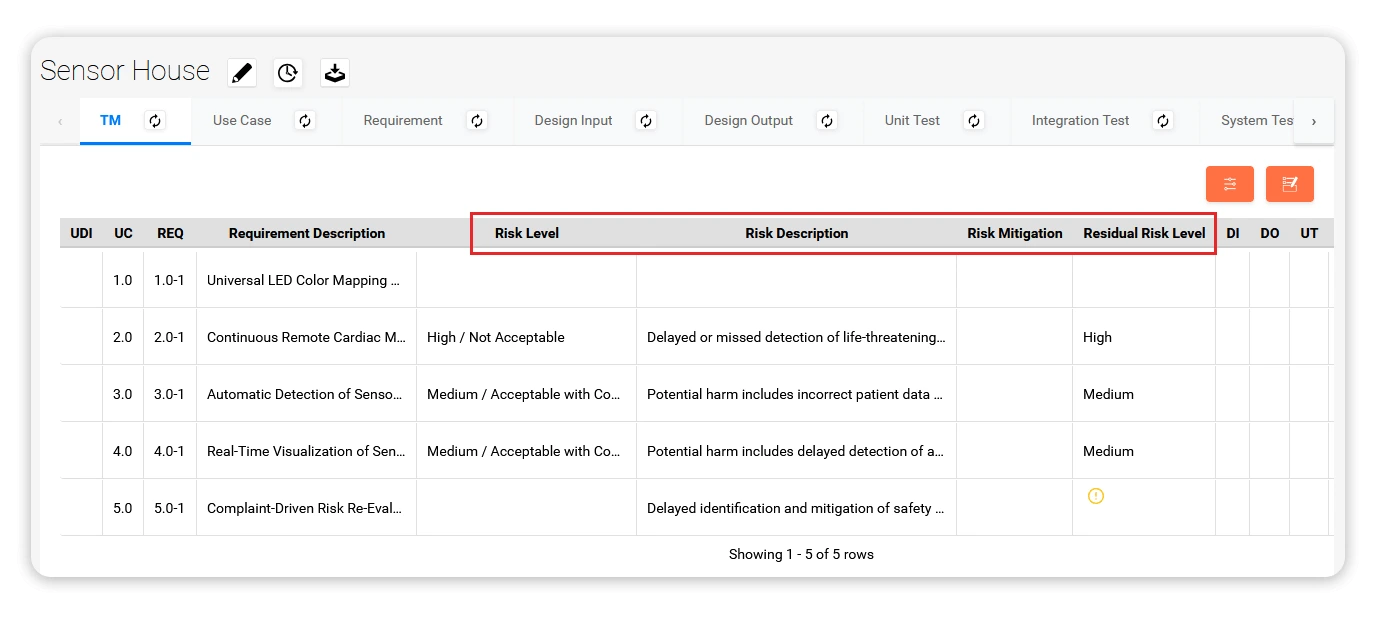
Aligned with design and traceability — without extra work
Risk records stay synchronized with Design Control and the Traceability Matrix.
As designs change and evidence grows, risk information stays consistent across the lifecycle.
Teams no longer need to:
- manually reconcile risk tables with design changes
- explain broken links during audits
- rebuild the Risk Management File from scratch
Clear visibility for teams and auditors
Dashboards and reports provide a simple view of:
- current risk status
- mitigated vs. open risks
- risks linked to CAPA or changes
This makes it easier to answer audit questions — and even easier to manage risk between audits.
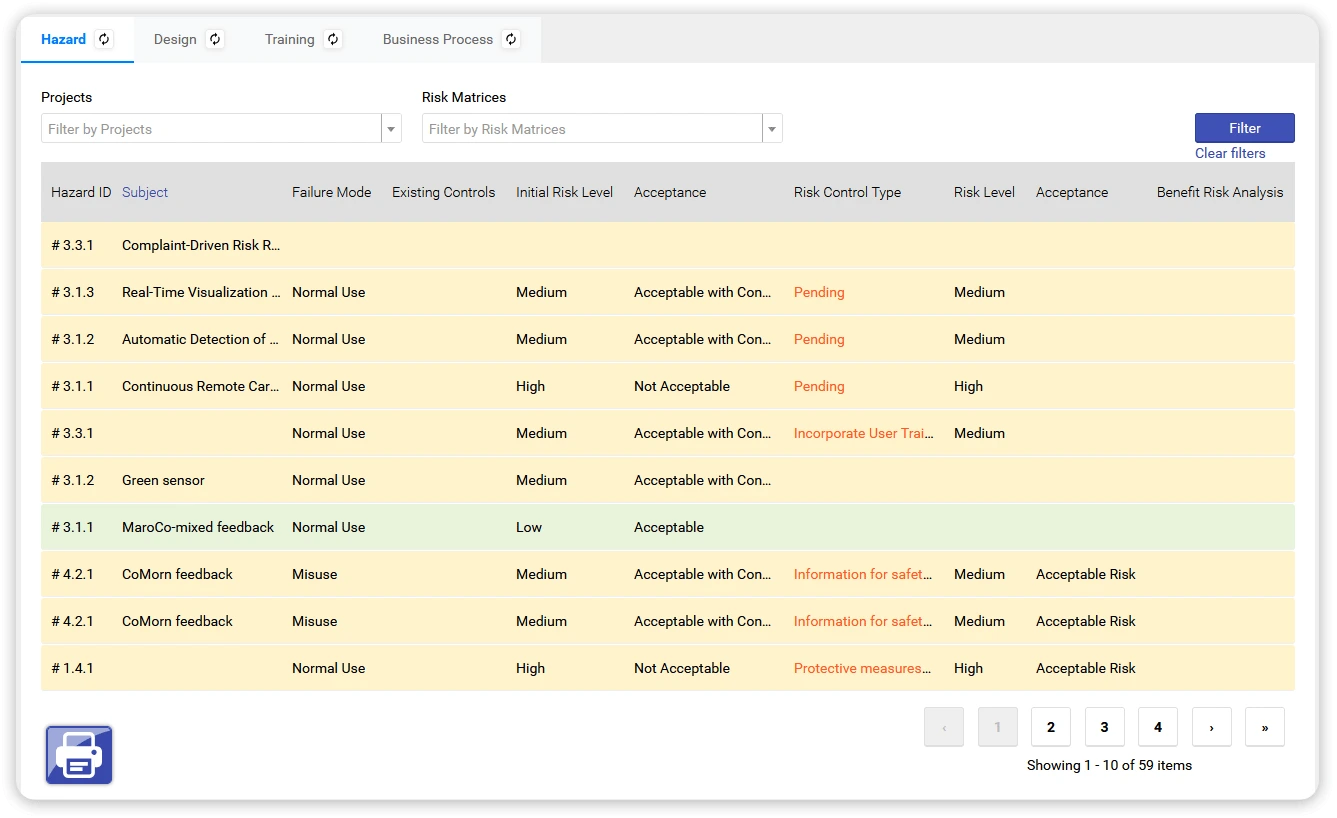
See how risk management can support your daily work — not slow it down
Understanding ISO 14971 Risk Management in Practice
What is ISO 14971 risk management for medical devices?
ISO 14971 risk management is the process of identifying, evaluating, controlling, and monitoring risks throughout the medical device lifecycle. It requires manufacturers to maintain documented evidence showing how hazards, hazardous situations, and risk controls are managed over time. In qmsWrapper, risk management stays connected to real quality and design activities instead of isolated spreadsheets or static documents.
What is a Hazard Log in a medical device QMS?
A Hazard Log is a structured record of hazards, hazardous situations, risk evaluations, mitigations, and residual risks associated with a medical device. In qmsWrapper, Hazard Logs are live and continuously updated as complaints, CAPA activities, design changes, or new evidence appear.
Why are live Hazard Logs better than static risk tables?
Static risk tables quickly become outdated because they rely on manual updates and disconnected documentation. Live Hazard Logs evolve together with the product and quality system, helping teams maintain accurate risk information without rebuilding the Risk Management File after every change.
How does qmsWrapper connect risk management to daily quality work?
qmsWrapper connects risks directly to requirements, complaints, deviations, CAPA activities, and design controls. This means risk is not treated as a separate activity performed only during audits or periodic reviews. Risk information stays connected to the workflows teams already manage every day.
What is event-driven risk re-evaluation?
Event-driven risk re-evaluation means risk is automatically revisited when meaningful quality events occur. Complaints, failures, deviations, or design changes can trigger risk reassessment immediately, helping MedTech teams react faster and avoid outdated assumptions.
How does automatic risk re-evaluation support ISO 14971 compliance?
ISO 14971 requires manufacturers to maintain ongoing risk management throughout the device lifecycle. Automatic risk re-evaluation helps ensure that new information affecting safety or performance is not missed, especially when complaints, CAPA actions, or design changes occur.
How does qmsWrapper support FDA QMSR expectations?
FDA QMSR moves toward a more system-based and risk-focused approach aligned with ISO 13485. qmsWrapper supports this by connecting risk management with quality events, traceability, CAPA processes, and design activities instead of treating risk documentation as isolated files.
How are complaints and CAPA activities connected to risk management?
In qmsWrapper, complaints, failures, deviations, and CAPA activities can automatically trigger risk reviews. This helps teams identify whether new issues affect existing risk assumptions, mitigations, or residual risk levels.
How does risk management connect to the Traceability Matrix?
The qmsWrapper Traceability Matrix connects risks with use cases, requirements, design inputs, design outputs, and testing activities. This creates full lifecycle traceability and helps teams understand how changes in one area affect related risk records and verification evidence.
Why is traceability important in ISO 14971 risk management?
Traceability helps demonstrate that risks, mitigations, requirements, and verification activities remain aligned throughout the product lifecycle. During audits, traceability makes it easier to explain how risk decisions were evaluated, controlled, and maintained over time.
How does qmsWrapper reduce manual risk management activities?
qmsWrapper reduces manual work by synchronizing Hazard Logs, CAPA workflows, design controls, and traceability records automatically. Teams no longer need to manually reconcile disconnected spreadsheets or rebuild risk documentation after every product change.
How does qmsWrapper help during audits?
Dashboards and connected records provide visibility into current risks, mitigated versus open risks, and risks connected to CAPA or changes. This helps teams answer audit questions faster and reduces last-minute audit preparation.
How does connected risk management improve product development?
Connected risk management helps teams understand how design changes, complaints, or verification results affect overall product risk. Instead of isolated reviews, risk stays aligned with ongoing product development and quality activities.
What is residual risk in medical device risk management?
Residual risk is the remaining risk after mitigation measures have been implemented. qmsWrapper allows teams to document residual risks, associated controls, and benefit-risk evaluations as part of a connected ISO 14971 workflow.
How does qmsWrapper support full lifecycle risk management?
qmsWrapper keeps risk records synchronized with design controls, CAPA activities, complaints, verification evidence, and traceability data throughout the entire medical device lifecycle. This helps maintain consistent and audit-ready risk documentation as products evolve.
