qmsWrapper 10.2 is now live, introducing advanced QMS change impact analysis alongside major improvements across AI, Change Management, Forms, Processes, and Technical File control.
At the center of this release is Wrapper-Mapper: an AI engine that analyzes change impact across your Technical File and the entire QMS.
Alongside this, the platform introduces:
- AI-driven risk analysis at the requirement level
- continuous traceability maintenance across the Technical File
- AI-assisted form completion and contextual input
- semantic AI search across the entire system
- a redesigned Forms engine and Process Editor
- integrated UDI tracking across workflows and documentation
This is not just a feature update.
It is a shift from passive QMS tools to active, operational systems.
Table of Contents
Wrapper-Mapper: QMS Change Impact Analysis Powered by AI
Quality management in medical devices is no longer about documentation.
It’s about understanding what changes, and why.
With this release, qmsWrapper introduces a new category of QMS:
an operational, AI-driven change management system that actively maintains your Technical File. At the center of this shift is Wrapper-Mapper, an intelligent engine designed to answer one critical question: When you make a change, what else across your QMS is affected?
The Problem: Change Is Documented — But Not Understood
Managing change in medical device development is rarely simple.
A small signal — a complaint, deviation, engineering observation, or feedback — can trigger a chain of consequences across the Technical File:
- Design inputs may need revision
- Risk assessments may require updates
- Verification protocols may need to change
- Training may be required
- Supplier documentation may be affected
In most organizations, identifying this impact still relies on manual investigation across disconnected systems.

The result:
- delayed decisions
- incomplete updates
- traceability gaps
- audit risk
Without proper QMS change impact analysis, teams struggle to understand the full scope of change before implementation begins.
The challenge is not documenting change.
The real challenge is understanding its full impact — before implementation begins.

If you want a deeper breakdown of how AI changes QMS workflows, download our AI QMS report.
The qmsWrapper Approach: From Event to Impact to Implementation
qmsWrapper is built as a connected system, not a collection of modules.
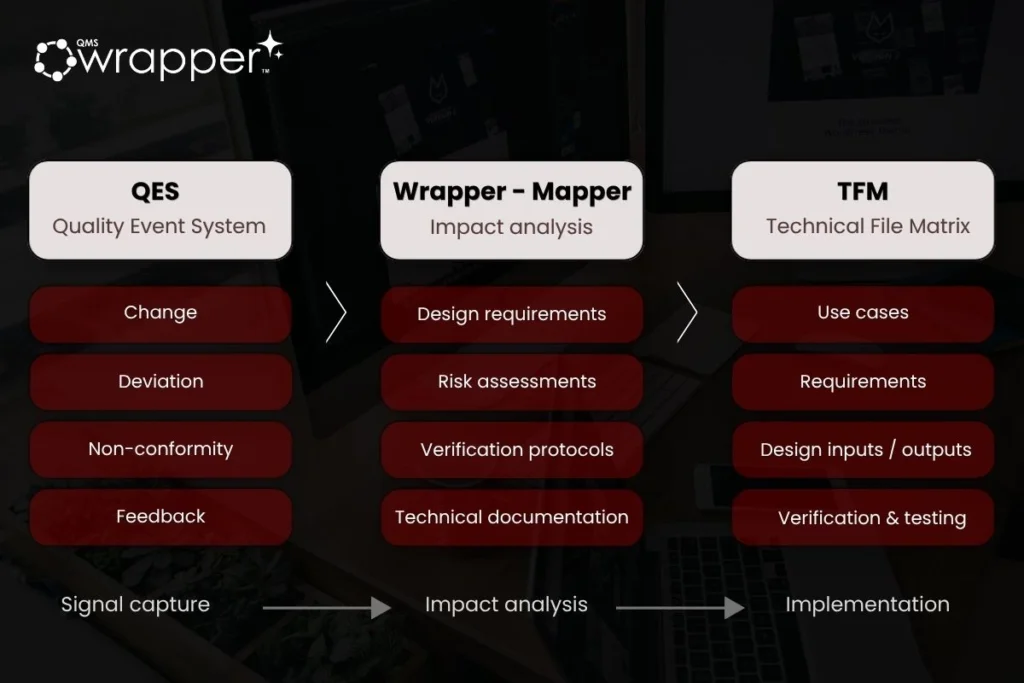
It operates through three core components:
- QES (Quality Event System) — captures signals
- Wrapper-Mapper — analyzes impact across requirements, risk, and the Technical File
- TFM (Technical File Matrix) — executes and maintains changes, including traceability and risk updates
Together, they create a continuous workflow:
Event → Impact Analysis → Implementation
This ensures every signal is:
- captured
- evaluated
- connected
- traceable
Capturing the Signal: Where Change Actually Begins
Change does not begin with a document.
It begins with a signal.
In qmsWrapper, all signals are captured through a structured QES Event Form:
- Change
- Deviation
- Non-conformity
- Feedback
Once submitted:
- the event is logged
- assigned automatically
- tracked in the Event Log
- visible to the QMS team immediately
This eliminates the most common failure point:
unstructured, lost, or delayed information.
Wrapper-Mapper: Change Impact Analysis, Redefined
Once an event is reviewed and approved, it moves to impact analysis.
This is where Wrapper-Mapper operates.
Wrapper-Mapper analyzes the event against the Technical File and identifies:
- affected requirements
- related risks
- impacted verification activities
- documentation dependencies
This is where QMS change impact analysis becomes operational — embedded directly into the workflow, not handled manually.
It then generates structured, actionable tasks describing what must be reviewed or updated.
The QMS team can:
- review
- refine
- regenerate
- approve
Once approved, tasks are automatically transferred into Change Management.
See how operational AI works in practice → download the AI QMS report.
Beyond Change Requests: Full Event Integration
Wrapper-Mapper is not limited to change requests.
Deviations, non-conformities, and feedback are routed through structured workflows:
- CAPA processes
- process improvements
- or linked change events
This ensures that every quality signal can lead to controlled, traceable change.
Technical File Execution: Fully Connected by Design
All approved changes are executed inside the Technical File Matrix (TFM).
The TFM structures the Technical File as a connected system:
- Use Cases
- Requirements
- Risk Assessments
- Design Inputs / Outputs
- Verification & Testing
Each element is linked.
Each update is traceable.
Each change is connected back to its origin.
This creates a continuous traceability chain:
Event → Decision → Change → Verification → Closure

AI That Works Inside the Workflow — Not Around It
In qmsWrapper, AI is not an add-on.
It is embedded into every operational step.
AI assists with:
- capturing and structuring events
- analyzing change impact
- generating follow-up tasks
- completing forms
- pre-filling risk assessments at the requirement level
- continuously maintaining traceability across the Technical File
- retrieving and connecting information across the system
Each module includes a dedicated AI assistant — but:
- all outputs are reviewable
- all actions are controlled
- all approvals remain human
This ensures:
efficiency without compromising auditability.
AI-Powered Forms: From Empty Fields to Guided Completion
Forms in qmsWrapper are now AI-aware.
Each field can include embedded AI prompts that guide the system to generate:
- context-specific suggestions
- relevant content
- structured inputs
FormAssistAI now:
- fills fields directly
- provides inline suggestions
- adapts based on previous data
Users no longer start from empty forms.
They start from structured, context-aware input.
AI Search: Finding What Matters — Instantly
qmsWrapper introduces semantic AI search across the entire system.
Users can search across:
- tasks
- forms
- risks
- documents
- chat messages
- processes
The system understands meaning — not just keywords.
Search results include:
- context
- relationships
- reasoning
This transforms search from retrieval into understanding.
UDI is Now the Fabric of qmsWrapper
Introducing UDI tracking at the project level, enabling UDI numbers to be linked to forms, tasks, processes, and related documentation across the system.
With this release, qmsWrapper embeds the Unique Device Identifier directly into the core of your QMS workflows—from Quality Events and Change Management through to the Technical File and Vigilance.
UDI now acts as the central thread connecting device data across approvals, traceability matrices, and AI-powered search. Whether managing a single device or a family of components, qmsWrapper ensures that UDI data is not only recorded, but actively integrated into the workflows that define and maintain your QMS.
Supporting Regulatory Traceability
Regulators do not evaluate documents in isolation.
They evaluate:
- decision logic
- traceability
- consistency
qmsWrapper ensures full visibility of:
- when a signal was detected
- how it was evaluated
- how risk was evaluated, documented, updated, and linked
- what was impacted
- how changes were implemented
This aligns directly with expectations under:
- ISO 13485
- ISO 14971
- FDA 21 CFR 820
- EU MDR
A Shift Toward Operational QMS
Most QMS platforms:
- store documents
- track records
qmsWrapper operates differently.
It connects:
- events
- decisions
- risk
- documentation
- execution
The result is a system that does not just record quality.
It actively maintains it.
From Reaction to Intelligence
QMS change impact analysis is one of the most critical capabilities in modern quality management systems.
But documenting change is not enough.
Understanding how change propagates across the Technical File is what defines audit readiness.
With qmsWrapper 10.2:
- signals are captured early
- impact is analyzed intelligently
- changes are executed in a connected system
- traceability is maintained automatically
From signal to impact.
From impact to implementation.
From documentation to intelligence.
Our Roadmap
Coming soon, as regular releases…
- Training Module — linking training to changes and CAPAs
- Supplier Management Module — integrating supplier-related workflows
- Technical File Master Log — a Dashboard and Log of all the Static files required in a TF by MDR and FDA.
- Vigilance Module — continuous monitoring of documentation
- Eudamed Module – A Dashboard and log of files submitted, deadlines, updates.
- Audit Module – Gap analysis, regulatory submission preparation tools…
Each module will integrate directly into the same event-driven architecture.
If you’d like to learn more about the 10.2 release or request a personalized walkthrough, please contact us at contact@qmswrapper.com or reach out to your account liaison directly.
Or download the AI QMS Report.
qmsWrapper
Less work. More control. Better audits.
Intelligence made operational.
Key Questions About Change Impact in Medical Device QMS
What is change impact analysis in a medical device QMS?
Change impact analysis in a medical device QMS is the process of identifying how a single change affects different elements of the Technical File, including design requirements, risk assessments, and verification activities. It ensures that all related documentation is reviewed and updated before implementation.
Why is change impact analysis critical for regulatory compliance?
Regulators do not assess documents in isolation. They evaluate how decisions are made and whether all affected elements have been considered. Change impact analysis ensures that organizations can demonstrate traceability, risk evaluation, and complete documentation updates during audits.
What makes change impact analysis difficult in traditional QMS systems?
In many systems, information is stored across disconnected modules. Quality teams must manually trace relationships between events, requirements, risk files, and verification records. This makes it difficult to understand the full impact of change before implementation begins.
How does Wrapper-Mapper improve change impact analysis?
Wrapper-Mapper analyzes events in relation to the structured Technical File and identifies which elements may be affected. It generates suggested actions for review and update, helping teams understand impact earlier and reducing manual investigation.
How does qmsWrapper ensure traceability across the Technical File?
qmsWrapper connects events, impact analysis, and documentation within a structured Technical File Matrix (TFM). This creates a continuous traceability chain from the initial signal to final implementation, making it easier to demonstrate compliance during audits.
Does AI replace human decision-making in change impact analysis?
No. AI in qmsWrapper acts as a support tool. It helps identify potential impact and suggests actions, but users remain responsible for reviewing, approving, and implementing all changes.
What role does the event-driven approach play in change impact analysis?
The event-driven architecture ensures that every signal — such as a change, deviation, or feedback — is captured early. This allows impact analysis to begin sooner, improving visibility and reducing the risk of missing critical dependencies.




