Event-Driven QMS
for Real-World Compliance
Every deviation, complaint, change, or audit finding becomes a structured, traceable record — connected to the right quality processes.
What “Event-Driven” Actually Means
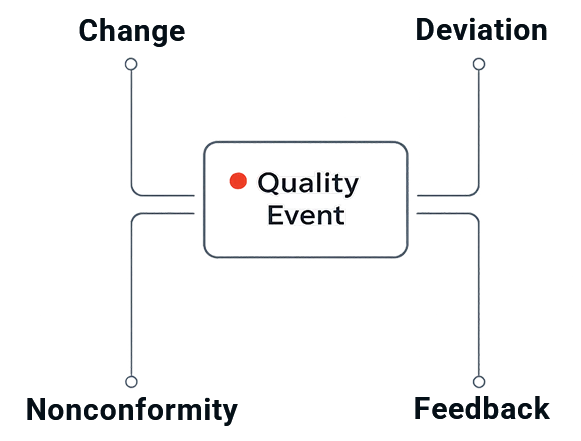
- Everything starts with a quality event
- Events are logged, tracked, and linked — not stored in silos
- Your QMS reflects what’s really happening, not just audit snapshots
This shift is exactly what FDA formalized with QMSR — moving inspections away from static procedures toward real-world system behavior.
👉 See how QMSR changes what auditors actually expect
An event is not the problem.
What happens next is.
No Over-Escalation.
No Missed Actions.
Not every issue needs a CAPA. qmsWrapper classifies events by type and impact — and guides the correct response.
- Minor issues → Tasks
- Serious issues → CAPA
- Design impact → Change Control
- Training gaps → Training actions
If everything becomes a CAPA,
nothing actually gets resolved.
Event-driven workflows keep focus where it matters.
From Capture to Closure — Fully Traceable
- Capture
- Assessment
- Action
- Verification
- Closure
Every step is timestamped, assigned, and auditable — nothing falls through the cracks.
Events Don’t Stop at Quality
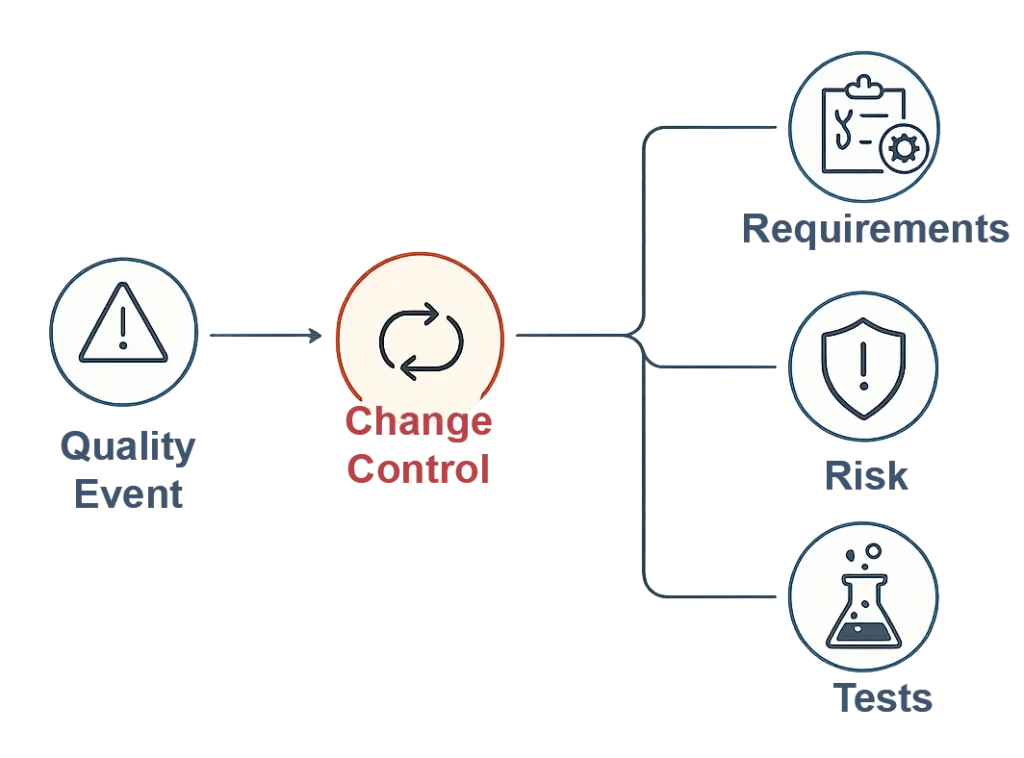
When an event affects risk or design, it can trigger Change Control — linking directly to requirements, risks, and tests.
- Closed-loop risk management
- Live traceability
- Audit-ready change history
Capturing events is only the first step.
Keeping everything connected after that is where most systems fail.
Complex processes don’t need complex behavior.
They need guidance.
AI-Assisted,
Not AI-Decided
- Guides users through complex QMS forms, step by step
- Suggests relevant fields, context, and linked records
- Helps complete forms faster — without skipping critical thinking
AI supports the process, while decisions, approvals, and accountability remain fully human.

Real-Time Visibility — No Audit Panic
- Open events & overdue actions
- Linked CAPAs and changes
- Clear status dashboards
Your logs, dashboards, and reports always reflect reality.
See Event-Driven QMS in Action
Events create the signal.
Understanding their impact is what makes your QMS effective.
Download the AI QMS report
Walk through real events, real workflows, and real audit trails.
Frequently Asked Questions about Event-Driven QMS
What is an event-driven QMS?
An event-driven QMS is a quality management system where real-world quality issues—like deviations, complaints, changes, and audit findings—are captured as structured records that automatically drive the correct workflow, actions, and audit trail.
What counts as a “quality event” in qmsWrapper?
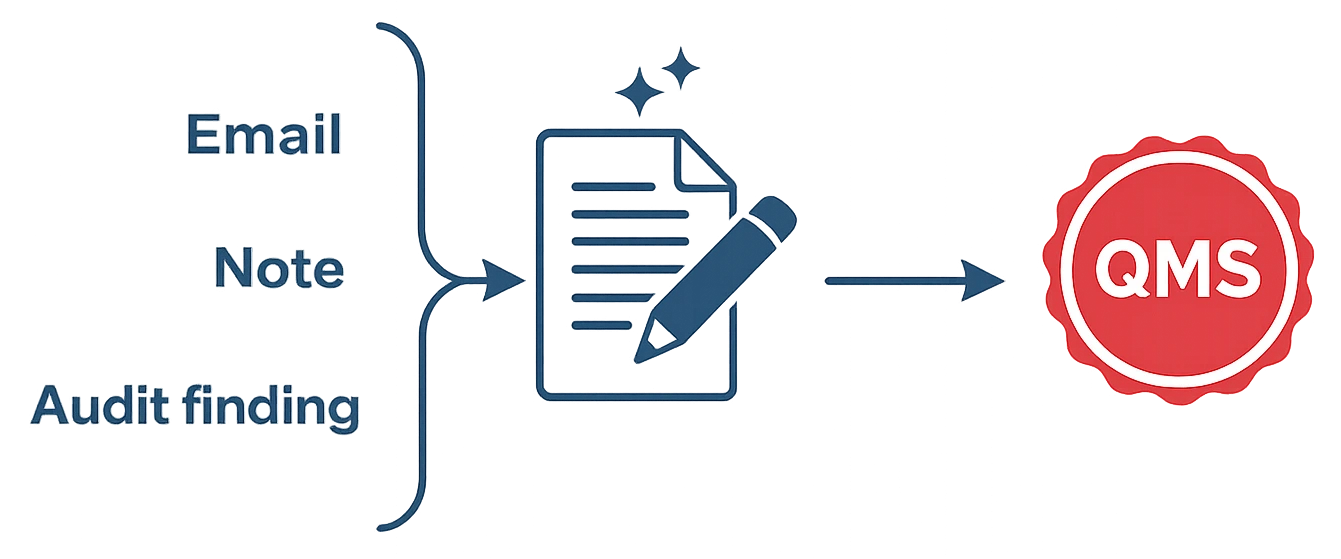
In qmsWrapper, a quality event is any signal that requires a controlled response, such as a deviation, nonconformance, complaint, audit finding, or change request—whether it starts from an email, note, meeting outcome, or formal report.
How is an event-driven QMS different from a traditional QMS?
Traditional QMS tools often reflect planned procedures and periodic audit preparation. An event-driven QMS reflects actual system behavior by continuously capturing issues as they happen and guiding users from assessment to closure with traceable evidence.
Does every event require a CAPA?
No. In qmsWrapper, events are classified by type and impact so the response matches the situation: minor issues become tasks, serious issues trigger CAPA, design-impact events trigger Change Control, and training-related root causes trigger training actions.
What is the lifecycle of an event in an event-driven QMS?
A typical event lifecycle is: Capture → Assessment → Action → Verification → Closure. Each step is assigned, timestamped, and auditable so nothing is lost and the full decision trail remains available.
What happens when an event impacts design or risk?
If an event affects design or risk, qmsWrapper can trigger Change Control and link the event to related requirements, risks, and tests—creating closed-loop risk management and an audit-ready change history.
How does event-driven QMS support FDA QMSR expectations?
QMSR emphasizes real-world system behavior and objective evidence across the lifecycle. An event-driven QMS supports this by showing how issues are captured, assessed, resolved, verified, and closed with traceable records—not just documented procedures.
What does “AI-assisted, not AI-decided” mean in qmsWrapper?
It means AI helps users complete complex QMS forms faster by suggesting fields, context, and relevant links, while all decisions, approvals, and accountability remain fully human and fully auditable.