Table of Contents
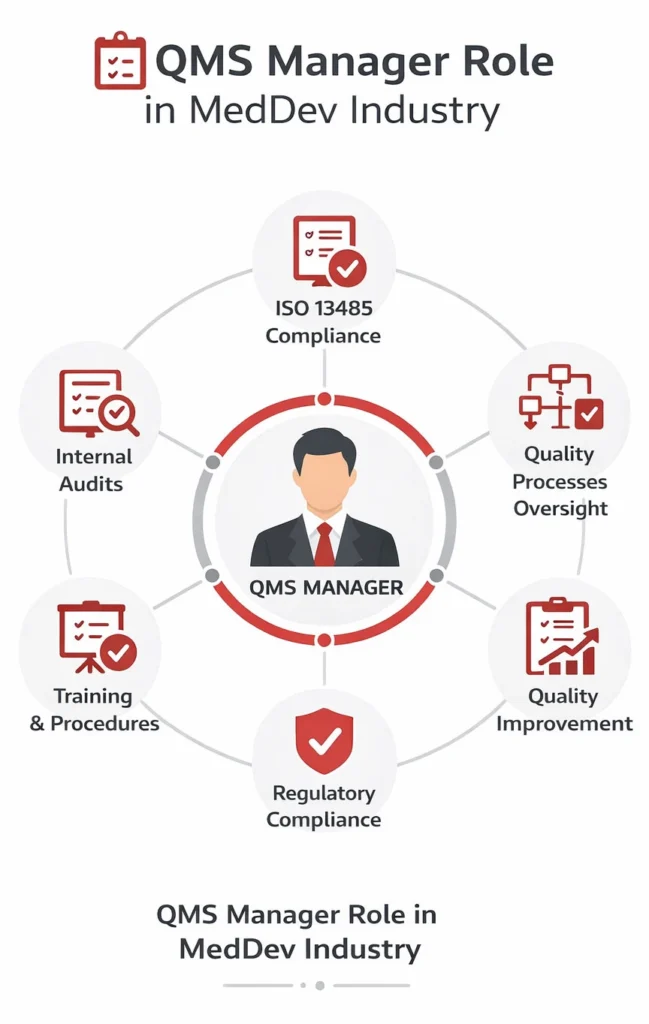
The QMS manager role in MedDev industry is critical for ensuring that quality management systems comply with standards such as ISO 13485 and support safe, effective medical device development. A QMS manager oversees quality processes across the organization, ensuring that procedures are followed and regulatory requirements are consistently met.
ISO 9001 sets out the criteria for a Quality Management System (QMS), a structured framework that ensures an organization’s products and services consistently meet customer and regulatory requirements.
It is expressed as the organizational goals, policies, processes, documented information and resources needed to implement and maintain it. It has four main components: quality planning, quality assurance, quality control, and quality improvement.
ISO 9001 specifies responsibilities for management in the quality management system, and it’s adapted to specific sectors and industries. It includes also ISO 13485.
ISO 13485:2016 specifies requirements for a quality management system where an organization needs to demonstrate its ability to provide medical devices and related services that consistently meet customer and applicable regulatory requirements.
Related to this: ISO 14971:2007 specifies a process for a manufacturer to identify the hazards associated with medical devices, including in vitro diagnostic (IVD) medical devices, to estimate and evaluate the associated risks, to control these risks, and to monitor the effectiveness of the controls.
In the medical device industry, ISO 13485 works together with ISO 14971 risk management requirements to ensure product safety and regulatory compliance. Within this framework, the QMS manager role in MedDev industry is responsible for ensuring that quality processes and risk management activities remain aligned with regulatory expectations.
QMS manager role in MedDev industry: focus on quality processes
Quality management focuses not only on product and service quality but also on the processes used to achieve it, using quality assurance and process control to ensure consistent and reliable outcomes.
It sounds like a tough and extensive job… No wonder it requires at least one person to deal with it!
QMS manager responsibilities in MedDev industry
A QMS manager safeguards that standards are met throughout all processes of the company, from customer relations to manufacturing processes and all in between.
A QMS manager is expected to provide instruction regarding company policies, standards, processes and practices and to ensure all employees follow company procedures in accordance with their role.
When it comes to the Medical Device Industry, the responsibility of top management is to ensure a QMS that is built in accordance with the structural requirements set forth in 13485:2016.
The responsibilities may include the following:
- Develop quality control processes
- Create and sustain the up-to-date product or process specifications
- Understand standards applicable to the business and ensure that they are met
- Train employees on quality assurance standards and processes
- Supervise the work of managers over various sectors
- Give instruction when standards are not met
- Check product development and manufacturing processes to ensure quality standards are met
- Identify quality improvement opportunities
- Provide quality reports
- Perform internal audits over the company’s quality systems
In practice, implementing the QMS manager role in MedDev industry requires tools that connect quality processes, documentation, and compliance activities in one system. Many medical device companies use specialized QMS software to support ISO 13485 processes, manage audits, and maintain full traceability across their quality management system.
If you want to see how an integrated MedTech QMS platform supports these responsibilities, you can book a demo of qmsWrapper to explore how quality processes, documentation, and compliance workflows can be managed in one system.
QMS manager requirements
To be qualified for a QMS manager position, it’s desirable to have a bachelor’s degree in a field related to quality control such as business administration or quality control management. Strong analytical skills to filter key points from complex details and develop plans accordingly and problem-solving skills are a must. They should be familiar with the methodologies, tools, and concepts of quality management. Also, they should have the ability to communicate the knowledge of industry standards to others. QMS managers should be able to interpret statistical data.
QMS managers need to have the following skills too:
- Effective communicators and excellent motivators
- Negotiating skills
- Report writing skills
- Excellent planning skills
Certain companies expect their QMS managers to have a thorough knowledge of the specifications that relate to their specific business. For such cases, they’re a lot of ISO training available. Even online…
QMS managers integrate their knowledge and expertise in order to accomplish set goals and always begin by first understanding and establishing their role and relationship with line staff. They play a crucial role in business by ensuring that products meet certain thresholds of acceptability. They play a critical role in ensuring that medical device companies maintain regulatory compliance while continuously improving quality processes.
In practice, the QMS manager role in MedDev industry is essential for maintaining regulatory compliance, managing ISO 13485 quality processes, and supporting safe medical device development.
Key Questions About the QMS Manager Role in MedDev Industry
What does the QMS manager role in MedDev industry involve?
The QMS manager role in MedDev industry involves overseeing the organization’s quality management system to ensure compliance with standards such as ISO 13485. A QMS manager coordinates quality processes, monitors regulatory compliance, manages internal audits, and supports continuous improvement across departments.
Why is the QMS manager role important in MedDev companies?
The QMS manager role in MedDev industry is critical because medical device companies operate in a highly regulated environment. A dedicated QMS manager ensures that processes follow regulatory requirements, quality procedures are implemented correctly, and documentation supports audits and product approvals.
What standards does a QMS manager typically work with?
In medical device companies, a QMS manager typically works with standards such as ISO 13485 for quality management systems and ISO 14971 for risk management. These standards define how companies must manage quality processes, documentation, and product safety throughout the device lifecycle.
What skills are required for a QMS manager in the MedDev industry?
A QMS manager needs strong analytical skills, knowledge of quality management methodologies, and a clear understanding of regulatory standards. Communication skills, process oversight, and the ability to coordinate quality activities across departments are also essential for success in this role.
Can the QMS manager role be combined with other roles?
In many organizations, especially startups, companies consider combining the QMS manager role with other positions. However, in the MedDev industry it is generally recommended to keep the role dedicated to ensure proper oversight of quality processes and regulatory compliance.




