Project and Quality Management for Medical Devices
Everything you need to manage work through projects — while quality and evidence are created automatically
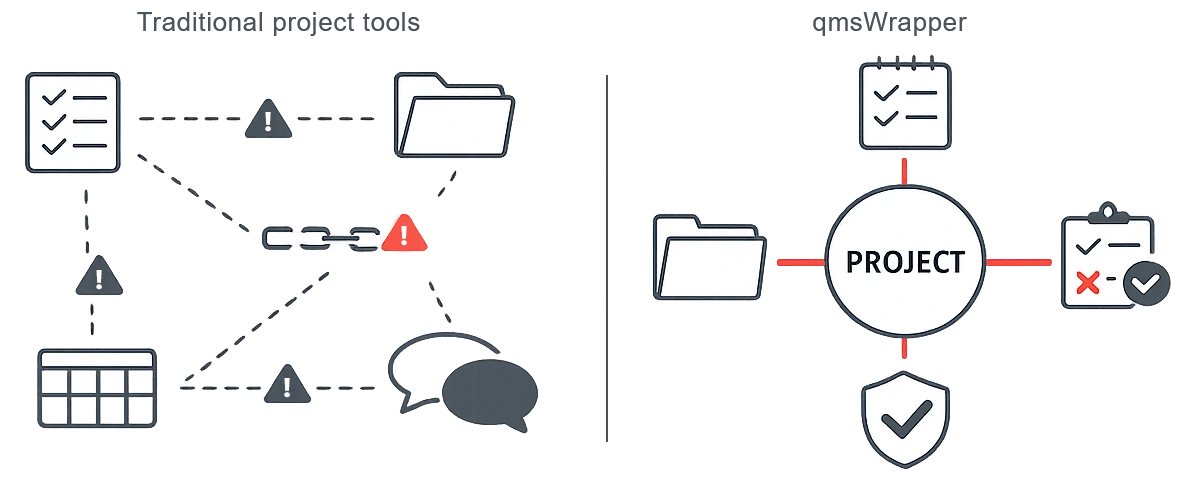
qmsWrapper combines Project Management & Quality Management into one system.
Projects are driven through defined QMS processes, so teams can move fast while quality oversight happens automatically — not as extra work.
Projects move.
Quality follows.
Evidence comes last.
In regulated environments, teams don’t fail because they work fast.
That leads to:
- work completed without full traceability
- changes approved without documented impact
- CAPAs, training, and follow-ups handled too late
These gaps usually surface only when auditors ask “why.”
The problem is not intent — it’s a structural misalignment between projects, processes, and evidence.
qmsWrapper closes that gap by managing work through quality.
If quality has to catch up later,
risk has already been introduced.
Audits don’t fail because teams worked fast — they fail because evidence was created too late.
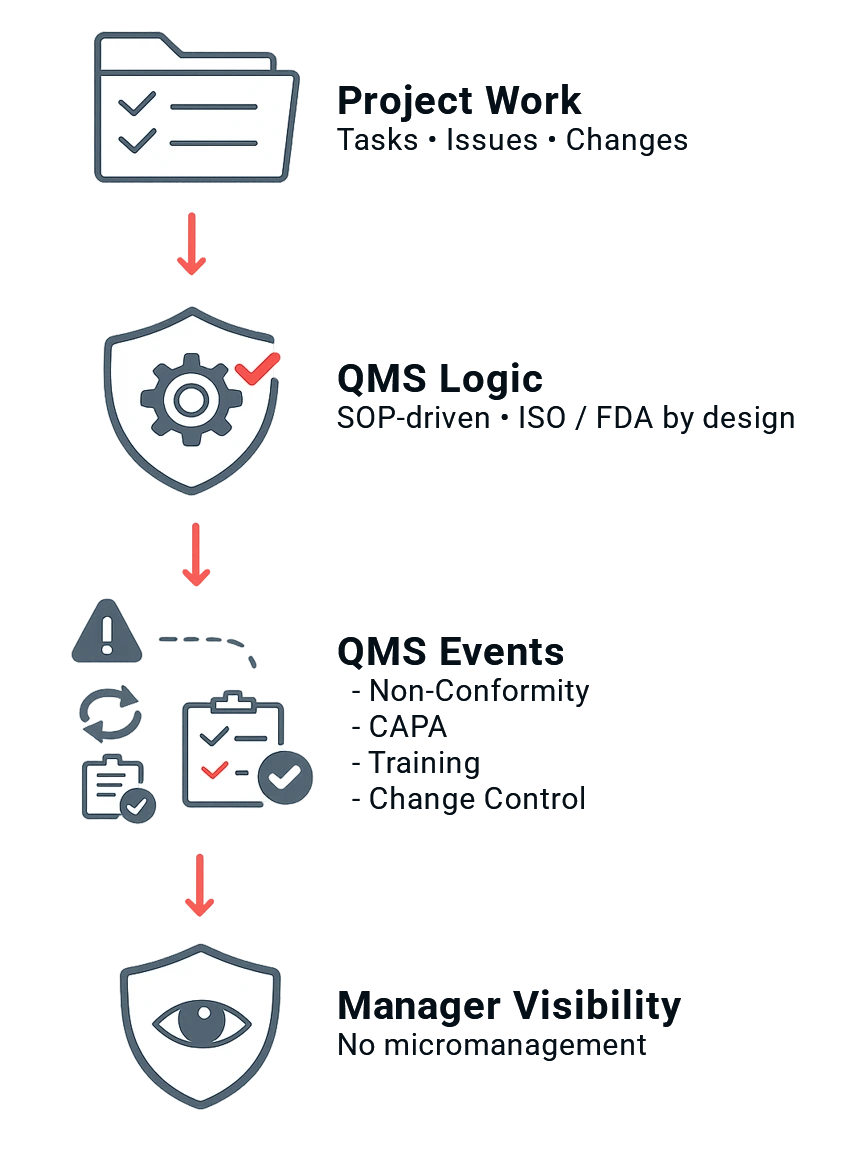
Managing Through Quality means:
Projects are not managed next to quality — they are managed through it.
As team members complete project tasks, the right QMS events are triggered automatically:
- non-conformities
- CAPAs
- audit findings
- training actions
- change controls
Managers get full visibility and control, without interfering with daily work and without adding another management layer.
Quality becomes part of execution — not something teams have to remember later.
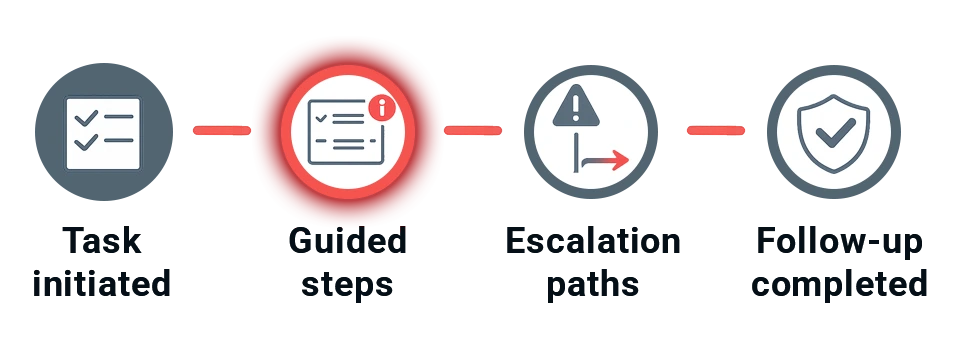
Step-by-step execution — without guessing what comes next
qmsWrapper uses defined QMS workflow processes to drive projects forward.
Each workflow provides:
- a clear sequence of steps
- built-in explanations and cautions
- predefined escalation paths
This creates a full circle of follow-up for every quality objective, task, and issue — with nothing left to guesswork.
ISO 13485 and FDA QMSR requirements are applied by design, not retroactively.
Your SOPs — automated
Your SOPs stop being documents — and start being executable workflows.
With the qmsWrapper Workflow Process Editor, you can…
- use ready-made ISO/FDA-aligned workflows
- adapt them to your own SOPs
- or build new processes from scratch in a few simple steps
Once defined, workflows automate the implementation of SOPs — ensuring consistency across teams and projects.
Reporting a non-conformity, managing a CAPA, or completing training becomes a natural part of project work.
One system, one view, one source of truth
From fragmented tools to coherent execution
Disconnected tools scatter evidence across project trackers, quality logs, documents, and emails.
qmsWrapper replaces that with one coherent, traceable view of what happened, why it happened, and how it was controlled.
When questions arise — internally or during audits — answers are immediate, consistent, and traceable back to execution.
Continuous audit readiness
Audit readiness is not achieved during audit preparation.
It emerges naturally when projects are executed through quality processes.
Evidence is created during work.
Approvals remain traceable.
Audits reflect reality.
Teams work normally.
Audits stop being disruptive.
Ready to manage projects where quality keeps up?
See how project work creates audit-ready evidence by design — without slowing teams down.
FAQ — Project and Quality Management for Medical Devices
What is project and quality management for medical devices?
Project and quality management for medical devices means executing project work through QMS workflows so that quality oversight, traceability, and audit evidence are created automatically during execution—not added later.
Why do traditional project management tools create audit risk in medical device companies?
Traditional project tools manage tasks and timelines but operate outside the QMS. This creates silos where work is completed without full traceability, changes lack documented impact, and quality actions are addressed too late—often discovered only during audits.
How does qmsWrapper manage projects through quality instead of next to quality?
qmsWrapper drives project execution through defined QMS processes. As tasks are completed, the system automatically triggers required quality events, ensuring projects and quality are managed as one integrated system.
Which quality events are triggered automatically during project execution?
During project work, qmsWrapper can automatically trigger non-conformities, CAPAs, audit findings, training actions, and change controls based on how tasks are executed and approved.
How does this approach support ISO 13485 and FDA QMSR compliance?
ISO 13485 and FDA QMSR requirements are applied by design through predefined workflows, escalation paths, and controlled approvals, ensuring compliance is embedded into daily work rather than handled retroactively.
How does qmsWrapper improve traceability across projects and quality activities?
All tasks, documents, approvals, and quality events are connected within a single project context, creating one coherent, traceable view of what happened, why it happened, and how it was controlled.
How does this model improve audit readiness?
Audit readiness emerges naturally because evidence is created during work, approvals remain traceable, and all actions can be traced back directly to execution—making audits a reflection of reality, not a reconstruction exercise.
Can SOPs be executed directly within project workflows?
Yes. SOPs are transformed into executable workflows that guide users step by step, ensuring consistent implementation across teams and projects without relying on manual interpretation.
Who should use project and quality management for medical devices?
This approach is designed for medical device and SaMD teams—especially startups and SMEs—that need to move fast on projects while maintaining continuous quality oversight and audit readiness.
