ISO 13485 QMS Software for Medical Device Companies
Manage CAPA, risk, design control, document control, and traceability in one connected system built for MedTech teams.
Pre-configured for ISO 13485, MDR, and FDA workflows.
Most teams go live in weeks, not months.
See how qmsWrapper connects QMS, DHF, risk, and change control in one audit-ready workflow.
Built for MedTech SMEs
Full traceability across QMS and DHF
No add-ons. One license
Why MedTech Teams Switch to qmsWrapper
Connected traceability
Link requirements, risk, CAPA, tests and changes automatically.
Built for MedTech SMEs
Structured workflows without enterprise complexity.
Fast implementation
Pre-configured system ready for regulated environments.
Trusted by MedTech teams in 25+ countries
qmsWrapper supports growing medical device and SaMD teams that need audit-ready quality processes without enterprise complexity.
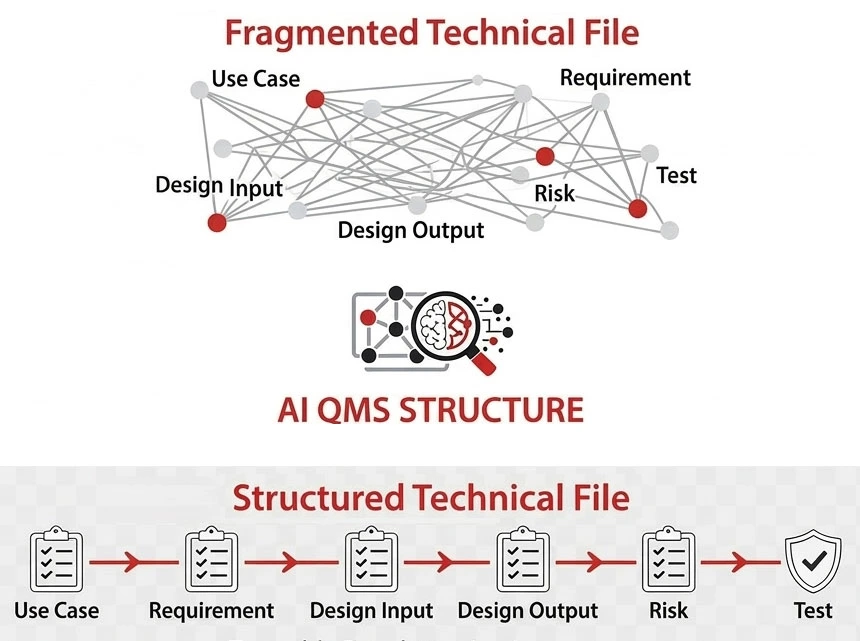
Why Many QMS Approaches Break as Teams Grow
| Paper-Based / Manual QMS | Traditional QMS Software | qmsWrapper | |
| Traceability | Manual and hard to maintain | Partly structured, often fragmented | Connected across QMS, DHF, risk, and change |
| Change impact | Manual analysis across multiple documents | Often requires manual cross-checking between modules | Related records stay visible across requirements, risk, and design data |
| Audit prep | Time-consuming | Better, but still document-heavy | Live audit-ready records |
| CAPA, risk, DHF | Separate files and folders | Often handled in separate modules | Structured and connected in one system |
| Team visibility | Limited | Partial | Shared real-time visibility |
| Daily work | Admin-heavy | Process exists, but can feel rigid | Guided workflows with AI-assisted support |
What works in the early stage often breaks when one issue touches CAPA, risk, validation, design, and documentation at the same time.

Implementation
Most MedTech teams start using qmsWrapper within weeks.
The platform is pre-configured for regulated workflows, so your team can begin structuring CAPA, risk, design control, and documentation without long configuration projects.
No complex customization.
No enterprise implementation cycles.
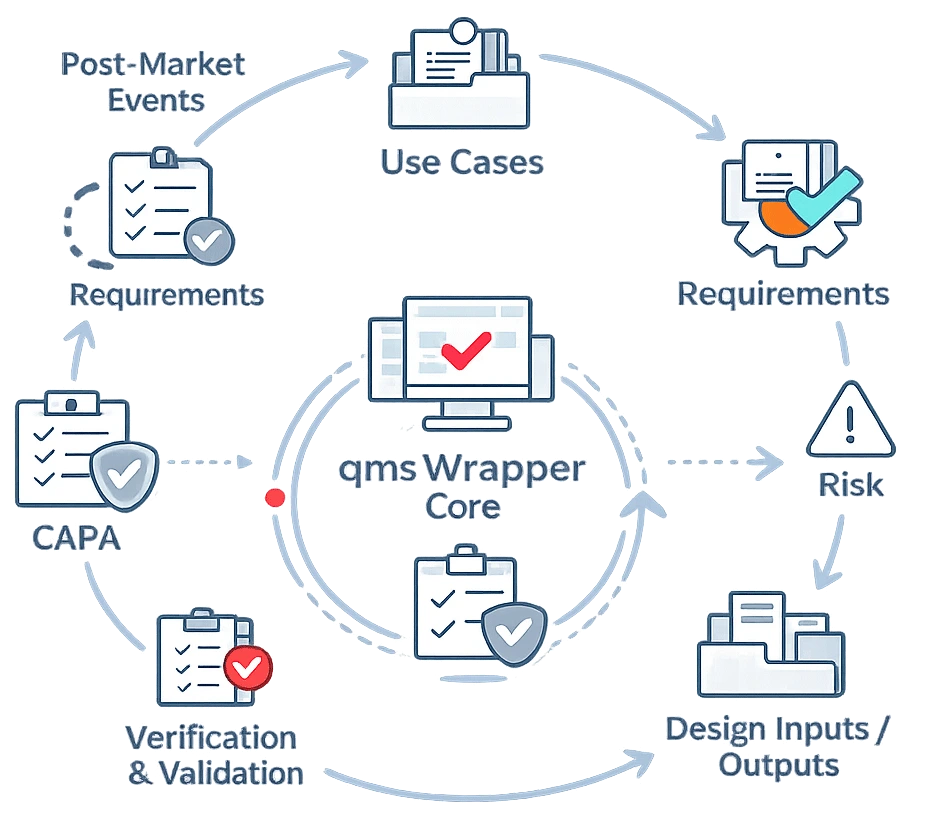
One Connected System for QMS, DHF, Risk, and Change
qmsWrapper gives MedTech teams one structured system where deviations, CAPAs, risks, requirements, tests, approvals, and documents stay linked automatically.
Instead of rebuilding the story before every audit, your team works in a live system of record from the start.
AI-assisted workflows help reduce manual data entry by pre-filling forms, transferring linked information between records, and suggesting relevant related items while keeping all decisions fully human.
Everything Your MedTech Team Needs in One License
- Event-Driven QMS for deviations, nonconformances, complaints, and change
- CAPA and root-cause workflows
- Design Control and Traceability Matrix
- ISO 14971 Risk Management
- Document Control and 21 CFR Part 11 eSignatures
- Audit and Supplier Management
- Training and Competence Records
- AI-assisted Smart Forms
No add-ons. No hidden modules. One connected system.
Built for MedTech SMEs — Not Enterprise Bureaucracy
qmsWrapper is designed for teams that need structure, speed, and traceability without long implementation projects or complex customization.
- Up and running in weeks
- Pre-configured for regulated workflows
- Easier adoption across quality, regulatory, and engineering teams
- Clear visibility from event to action to closure
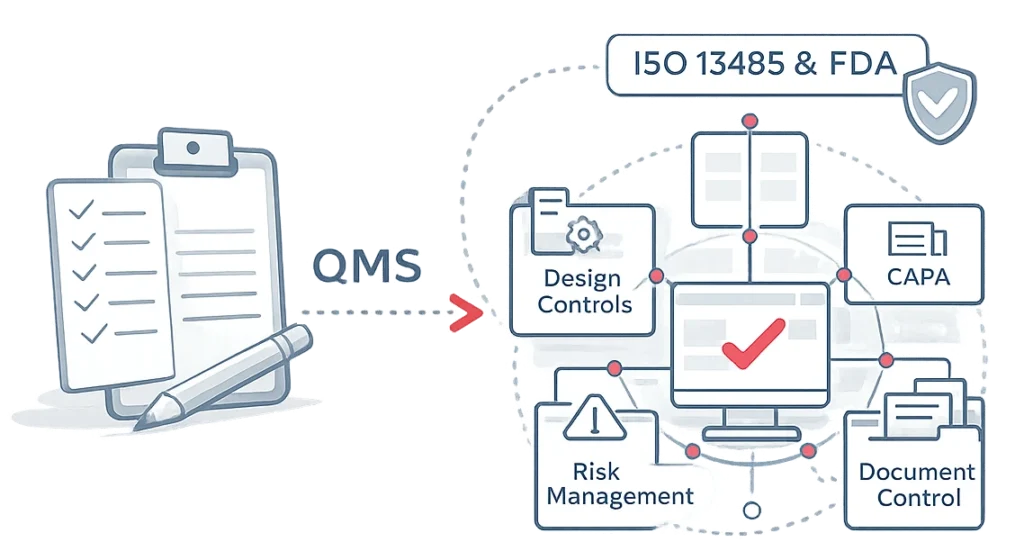




















Pre-Validated and Ready for Regulated Environments
qmsWrapper supports teams working under:
- ISO 13485
- ISO 14971
- FDA 21 CFR Part 820
- FDA 21 CFR Part 11
- EU MDR and IVDR
- UK MDR
- SaMD and regulated software environments
With a single system of record, every change, deviation, and CAPA
stays connected and audit-ready.
What You’ll See in the Live Demo
During the live demo, we’ll show you:
- how CAPA, change, and risk stay connected
- how traceability links requirements, tests, and DHF records
- how AI-assisted forms reduce manual work
- how qmsWrapper supports faster audit preparation
- how quickly your team can get started
No generic sales pitch. Just a practical walkthrough of workflows MedTech teams use every day.
Prefer to book directly? Schedule a time here.
Schedule Your Personalized qmsWrapper Demo
See how a Medical Device QMS works in real regulatory scenarios.
Testimonials
What our clients said
“Being a small start-up company, having ISO 134185 to maintain and CE and FDA pending, we are quite happy how we use this software. The custom forms help us to completely transfer our work there instead of having to go back and forth between Word and software.”
Alex Hayes
(CTO, Inova Medical)
“As a startup we were looking for a solution that’s cost-effective to implement, with the functionality we need now, but that can scale up as our needs become more complex. qmsWrapper can grow with us.”



















